Heart rate variability monitoring with wearable solutions
Update: Nov 4th 2025
This blog post provides an overview of the application of heart rate variability (HRV) for monitoring purposes - especially with wearable solutions. HRV operationalizes the successive beat-to-beat fluctuations over a defined period of time, is derived from the time series of successive R-R intervals using various context-dependent metrics, and reflects the complex dynamic modulation of the heart’s chronotropic response to physiological and/or pathological perturbations. HRV metrics are used as markers of human cardiovascular health and risk stratification, or as measures of load quantification, exercise response and performance, respectively. However, a valid use of HRV in the fields of sports medicine and exercise science requires careful consideration of the specific measurement principle of the recording device, standardized assessment, preprocessing, analysis, and context-sensitive interpretation. You can find further insights and references in our "Clinical Review" for the German Journal of Sports Medicine or in the bibliography section below.
Background of heart rate variability
Heart rate (HR) variability (HRV) describes the temporal change (variation) of the heart period or beat-to-beat variability of the HR over a defined measurement period and refl¬ects the dynamic end-organ response of the heart to physiologic and/or pathologic perturbations. The time series of successive R-R intervals is called „tachogram“ from which various metrics with different durations (e.g., from ultra short-term (1 min), short-term (5min) up to long-term (24 h) depending on the context) are derived. HRV is primarily created by the non-linear interaction of the efferent positive chronotropic in¬uence of the sympathetic (SNS, „fight or fl¬ight“ response), the negative chronotropic infl¬uence of the parasympathetic (PNS, „rest or digest” response) branch of the autonomic nervous system (ANS), and the intrinsic activity of the hearts pacemaker cells and refl¬ects the context-dependent psycho-neuro-endocrinological modulation of cardiovascular control. This complex modulation integrates several feedforward and feedback mechanisms that act on different time scales (e.g., central command, arterial barore¬flex, respiratory control, circulating catecholamines, muscle metabo-/mechanore¬flex, renin-angiotensin-aldosteron-system) and is therefore related to the dynamic regulation of circulatory control of arterial blood pressure and HR to meet the context-dependent needs of the whole organism. Thus, different evidence-based HRV metrics are used as markers of human cardiovascular health and risk stratification or as measures of load quantification, exercise response and performance, respectively. However, for a valid use of HRV in the fields of sports medicine and exercise science careful considerations on standardized assessment, preprocessing, analysis, and context-sensitive interpretation are mandatory.
Assessment and standardization
A valid and reliable assessment of the time between two successive heartbeats, referred to as the R-R interval in milliseconds, is most crucial for a scientific and practical use of HRV as the R-R interval (based on the detection of the R-wave in the PQRST complex of an electrocardiogram, ECG) time series serves as the base for any HRV metric.
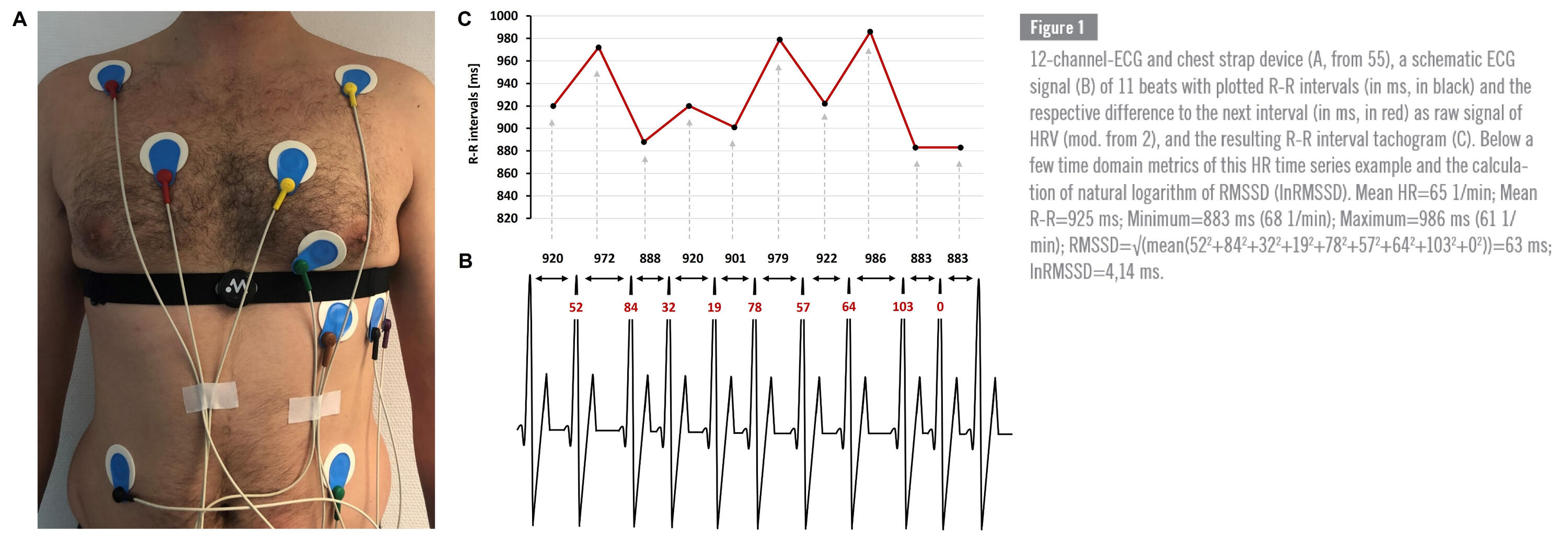
The gold-standard for R-R assessment is the ECG, which allows recordings in the laboratory or during daily activity (up to
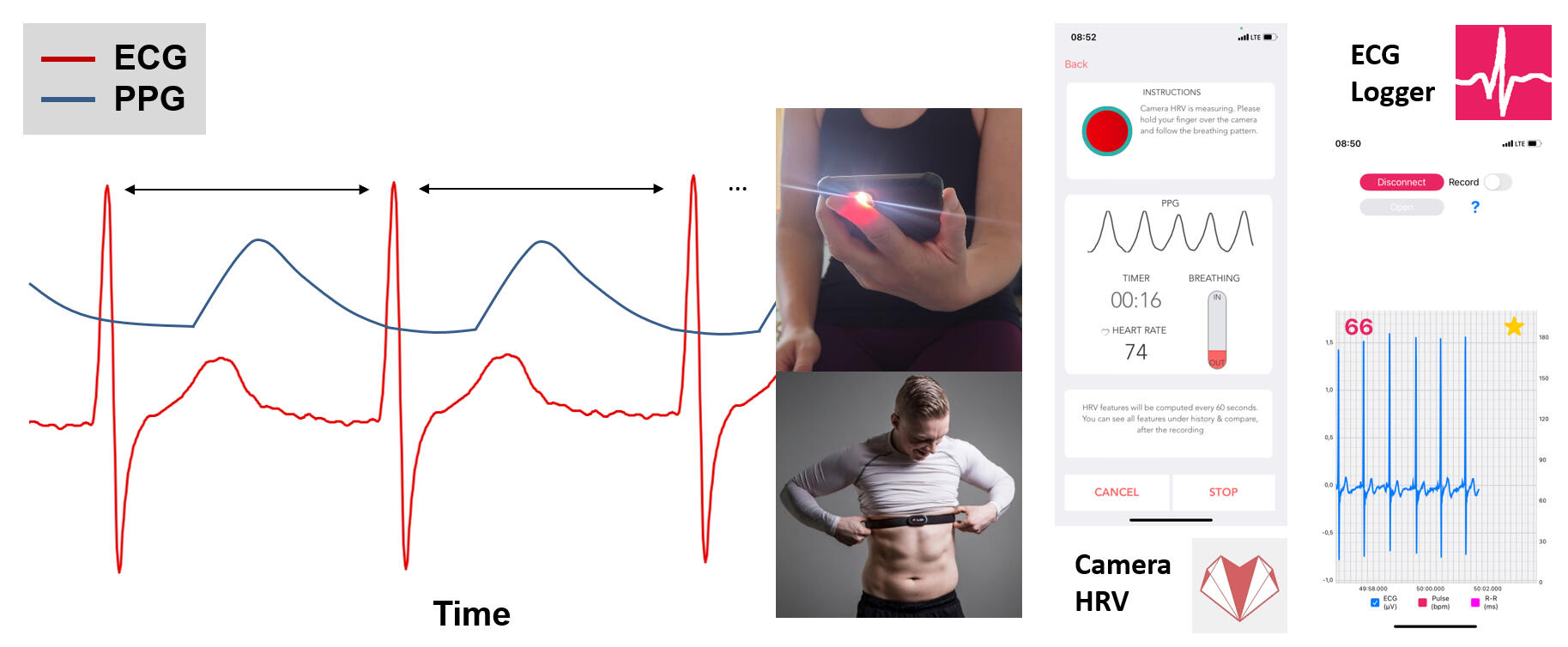
24 hours or longer). In addition, several mobile and user-friendly measurement systems and wearables exist, which can record R-R intervals with different applications and form factors (chest strap systems, adhesive patches and electrode systems on the skin) that also utilize electrophysiological, but also optically derived signals such as photoplethysmography (PPG).For valid HRV acquisition through electrophysiological measurements, a sampling rate of at least 250 Hz is recommended to accurately detect the R-peak. Specific analyses show robust results even at lower sampling rate depending on the selected HRV analysis parameters and principle of measurement. In order to increase usability and user compliance in field applications, new measurement principles based on PPG are now being used also for HRV assessment during resting conditions. PPG is a simple non-invasive optical technique in which the skin is illuminated by an LED (for example, the flash of a phone) and the reflected amount of light is detected by a photodetector or camera near the light source. The amount of reflected light depends on the volume in the vascular system. After systole the blood volume increases, decreasing the received light intensity, and during diastole the blood volume decreases, increasing the light reflection - therefore, the volume changes can be used to detect the pulse wave. The PPG signal can be used to detect HR and HRV - but more specifically, the signal provides pulse rate (PR) and pulse rate variability (PRV).Below you can see one schematic example of a single lead ECG tracing and the pulse wave from PPG technology as well as two smartphone applications to collect the PPG signal via the phone camera and ECG signal with a chest strap device.
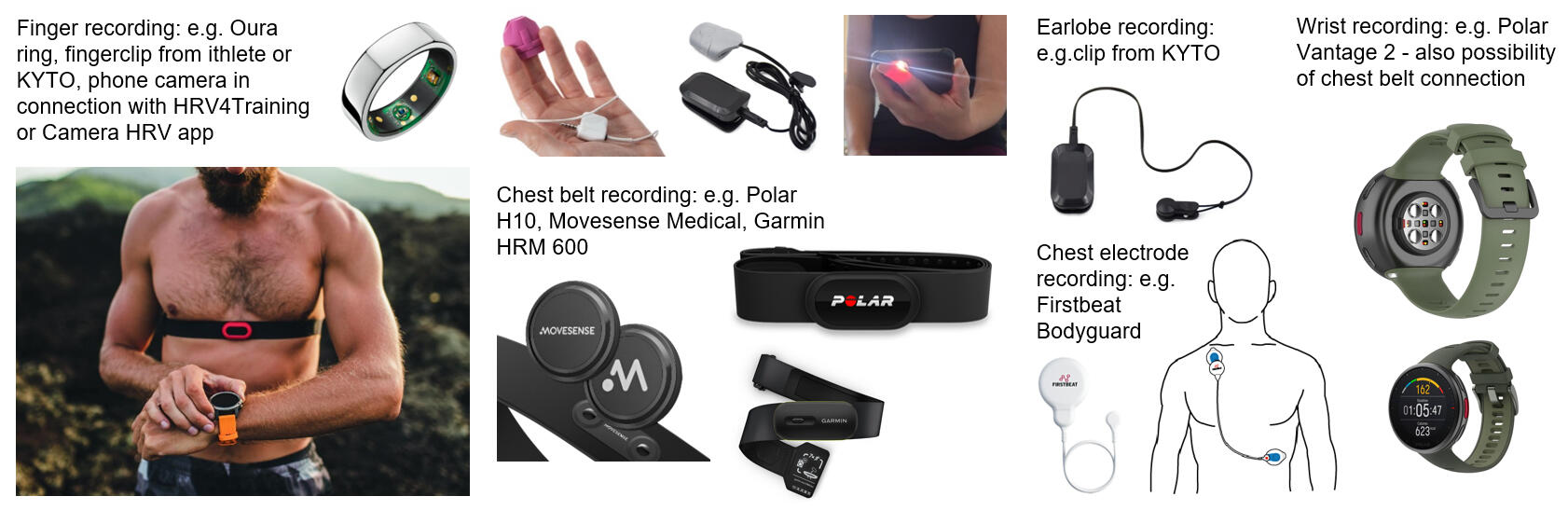
Most commercially available portable devices show a low absolute error under resting conditions, but should always be validated against reference measures to clarify the accuracy of data parameters and maximize real word application value. It is essential to evaluate the validity depending on the setting, measurement duration, paradigm and cohort that is investigated, because modulators such as the analyzed metric, body position, or individual characteristics of the population can cause deviations in HRV measurements from different devices. For applied settings, different portable high-resolution systems have been validated such as chest strap sensor systems for ECG-accurate electrophysiological recordings coupled via Bluetooth connection with receiving devices (smartwatches, smartphone via apps) for acquisition during rest and physical exercise conditions. Finally, PPG short-term measurements (metric: RMSSD) with the index finger and smartphone camera (light signal: flashing light) can currently also be recommended depending on the specific solution under resting conditions. Other PPG technique form factors like finger rings present promising user-friendly applications for frequent measurements in standardized measuring intervals, e.g., during the night.
A clear standardization procedure is essential to avoid and/or correct artifact in collected HRV data for research and practical application (36, 54). This is especially true for ultra-short-term measurements. A non-exhaustive list of potential artifact causes and influencing factors (acute and chronic) depending on the measurement principle can be found below.
Hardware and physiological considerations:
Ectopic beats, atrial fibrillation
Electrophysiological signal strength
Measurement site of the application
Sensor or electrode geometry and contact (pressure)
Wavelength of light
Transmission of the light reflection
Software considerations:
Sampling rate
R-wave detection algorithm
Preprocessing of data (e.g., detrending method)
Artefact correction incl. digital filtering (e.g., electrophysiological and motion artifacts)
Algorithms and default settings of signal processing (e.g., frequency bands)
Considerations of measurement protocol incl. personal and environmental factors:
Resting state or stress / exercise conditions (e.g., intensity, duration, modality)
Time of day (e.g., morning vs. night) and seasonal effects
Body position (e.g., lying vs. standing)
Duration of measurement and chosen HRV metric (time, frequency or non-linear domain)
Environment (e.g., temperature, humidity, sun and light exposure, hypoxia)
Age, sex and genetics
Hormone status (e.g., menstrual cycle, pregnancy)
Caffeine and alcohol intake
Medications
Bladder filling and hydration status
Nutrition intake
Health status (e.g., chronic disease, acute viral infection, food poisoning)
Acute and chronic stressors (e.g., occupation, travel, time shift and jetlag)
Physical activity and performance level
Body composition
Exercise load (e.g., intensity, duration) and recovery status
Injuries (e.g., impaired mobility) and pain status
Sleep quality and duration
Breathing pattern (e.g., paced breathing vs. natural rhythm and free running system)
HRV analysis
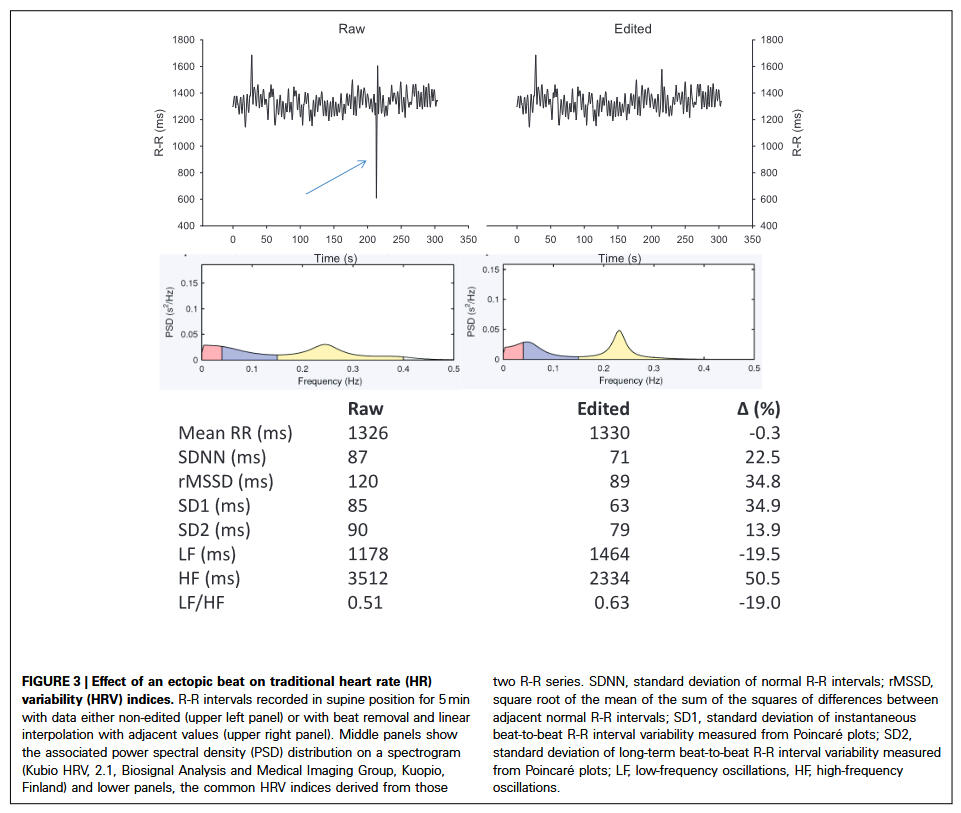
All HRV assessment technologies are affected by artifacts and/or noise, therefore disclosure of artifact detection/correction methods as well artifact rates (mostly below 5-10%) is mandatory. This is especially true for applied settings that involve different body positions, body movements and physiological strain. Besides the context-sensitive choice of measurement duration, as well as the detection and removal of artifacts (e.g., extra beats, noise, missing beats), HRV analysis software applications should include detrending and interpolation options for R-R time series using transparent algorithms to ensure sufficient data quality and to fulfill signal requirements for further calculation of specific metrics of the time-, frequency-, and non-linear domains (see table below). The following figure and caption shows the influence of one single ectopic beat artefact on the visual signal and HRV metrics.
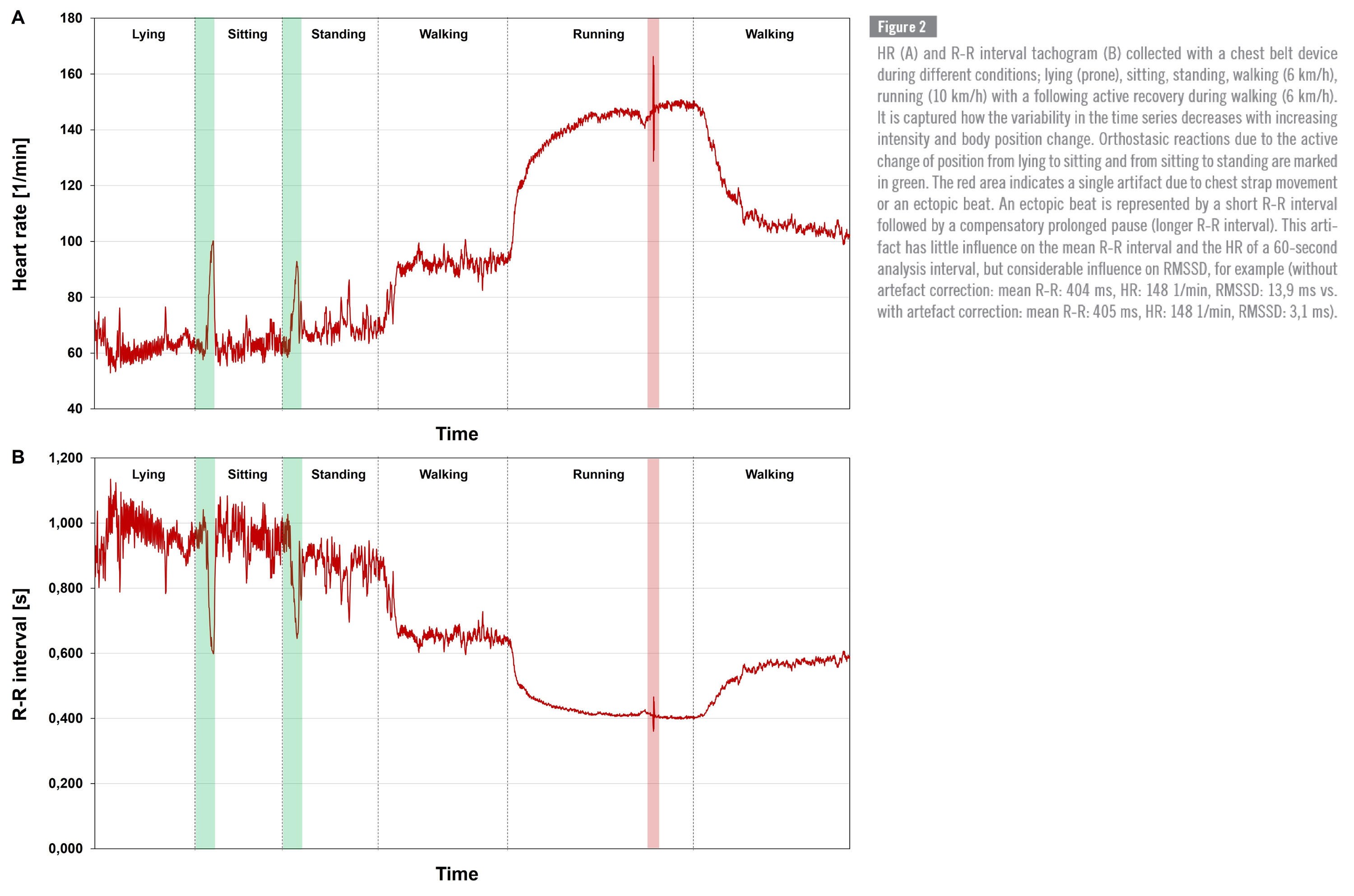
In addition, in an overview from Martin Buchheit about monitoring of HR measures a further example is illustrated about the influence of artifact influence on HRV metrics. Below is a tachogram showing artifacts and a clean part of the signal to give an impression.
Due to the continuous technical development of measurement systems, applications, and measurement principles, current validation studies should be consulted in order to assess the validity and quality of recording (and analysis) for the specific measurement context. Below are two reviews presented comparing different applications and measurement principles currently available on the market.
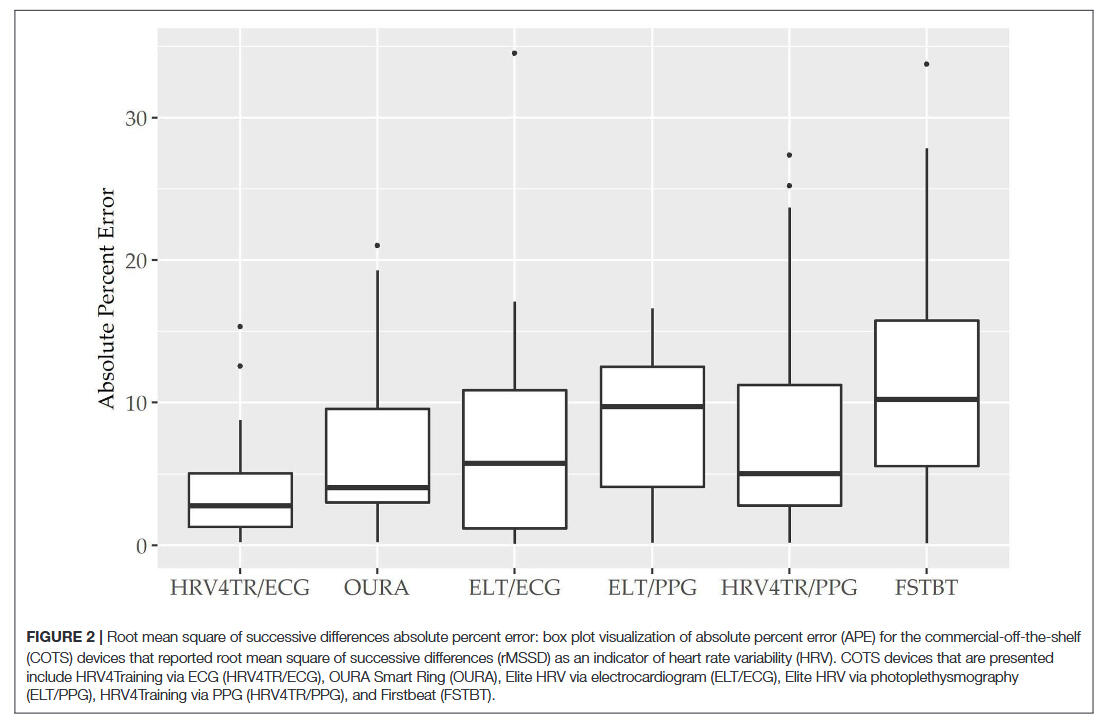
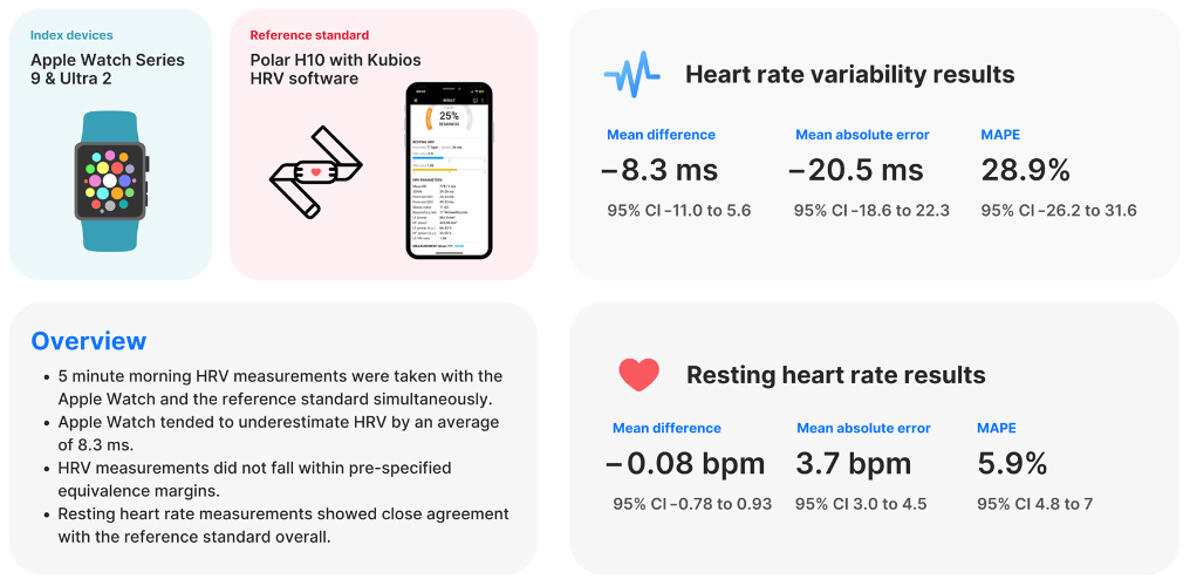
The results suggest that HRV measurements acquired using portable devices demonstrate a small amount of absolute error when compared to ECG. However, this small error is acceptable when considering the improved practicality and compliance of HRV measurements acquired through portable devices in the field setting. Practitioners and researchers should consider the cost-benefit along with the simplicity of the measurement when attempting to increase compliance in acquiring HRV measurements.However, these rather general statements do not exempt the specific evaluation of a new measuring device. You can find one example for the Apple Watch Series 9 with the considerable width of possible measurement errors to be taken into account for HRV measurements. However, it is difficult to keep up with the rapid pace of technological development in order to provide constantly updated validations for different application scenarios and populations.
For the scientific use of automatically calculated HRV metrics in commercially available systems and applications at least raw data access and sufficient information on the above-mentioned preprocessing features as well as the calculated metrics itself should be provided. Composite scores (see below for an overview) of different HRV metrics and other included data sources or estimated features without further information should be treated with caution. As mentioned above when analyzing HRV, reference should always be made to specific HRV metrics of the time-, frequency-, and/or non-linear domains, as these metrics may capture quite different HRV components, and their interpretation is always context-sensitive and depends on the setting.Overview of composite scores related to HR and HRV measurements:

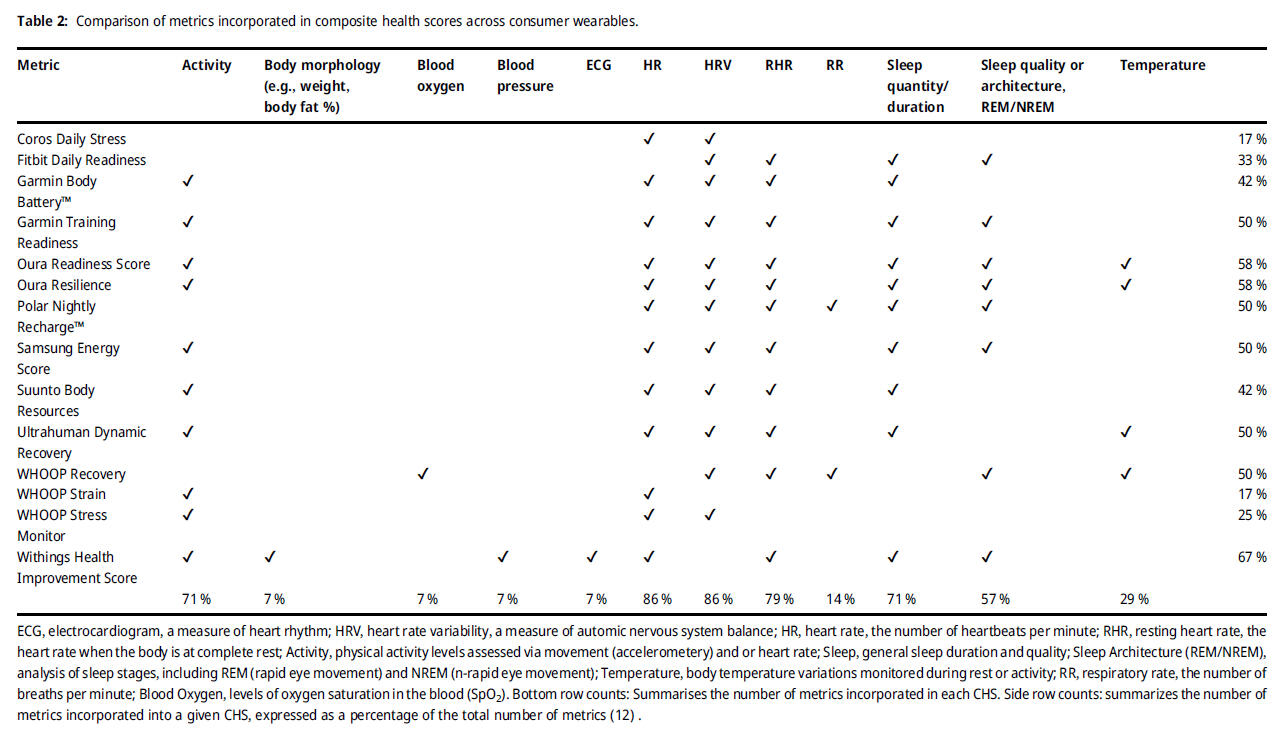
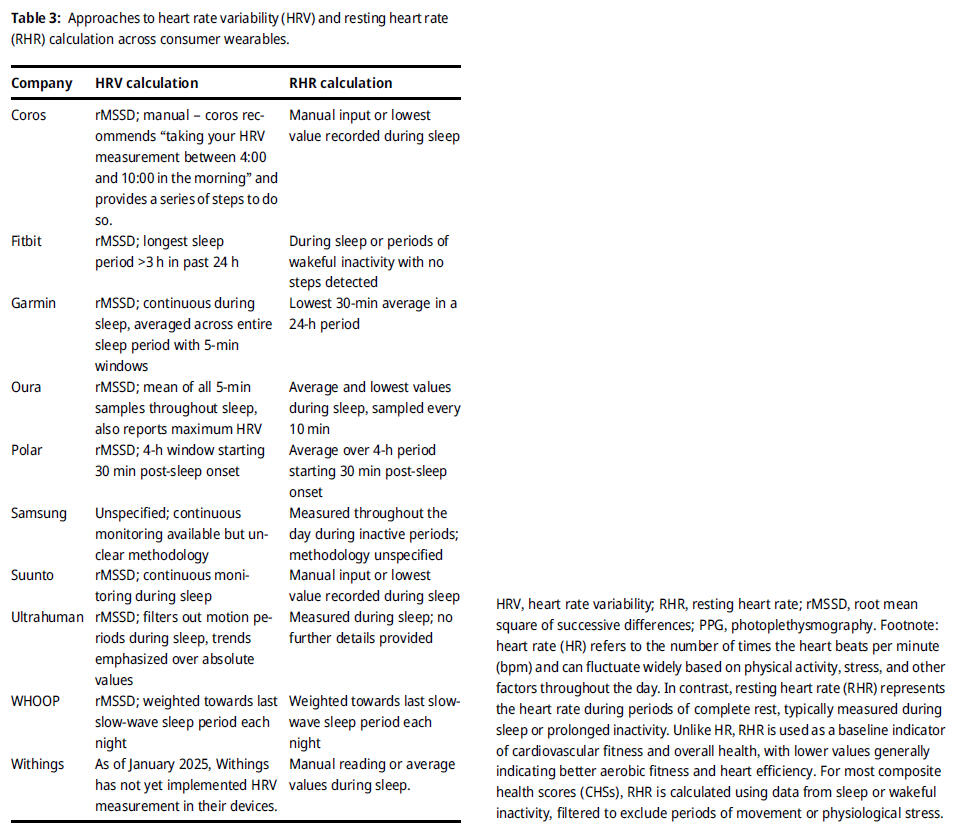
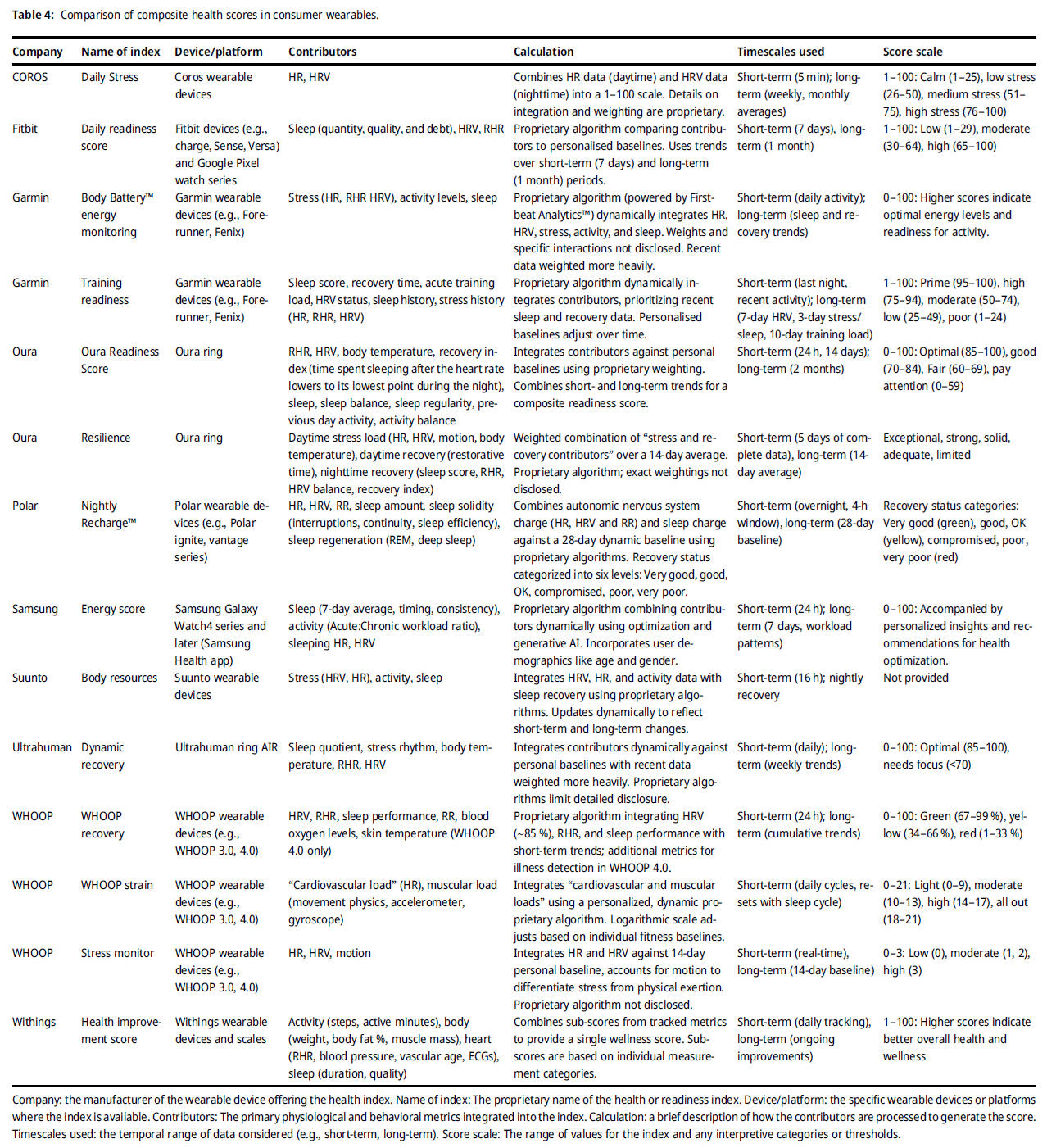
Further, „Upside Analysis“ provides a recent overview about the HRV vendor ecosystem market with key trends, challenges, case studies, and recommendations (click on the picture):
Coming back to standards of HRV analysis, most common linear time- and frequency-domain HRV metrics display rather general descriptive statistical features or display the distribution of the frequency content of the signal. For example, the standard deviation of all normal-to-normal R-R intervals over a given time interval (SDNN) is a general estimate of the global variability of the time series. In addition, the amount of efferent vagal modulation can be estimated from the several parasympathetic-dominated HRV metrics like root mean square of successive differences of NN intervals (RMSSD), high-frequency (HF) Power, or SD1 from the Poincaré Plot, while metrics from different recording durations may not be used interchangeably and their physiological meaning may vary considerably.
Although 24h HRV-recordings used to represent the “gold standard” for clinical HRV assessment, short-term and ultra-short-term measurements are increasingly used and validated for specific applied settings. Further, it should be noted that for several HRV metrics there is still an ongoing debate about their practical relevance and optimal recording lengths as these strongly depend on the algorithm and the setting. E.g., for stable frequency domain metrics, a recording length of two to ten times the wavelength of the lowest relevant frequency component is recommended, while this might be partly overcome by more sophisticated time-frequency analysis. In addition, several other HRV metrics from e.g., the geometric domain (e.g., HRV triangular index, TINN) were introduced for specific settings, but are not frequently used in sports medicine and exercise science. Therefore, the information from the table above should only be seen as a guideline for a large variety of applications in the field of sports medicine and exercise science.In addition, R-R ¬fluctuations from short term recordings like RMSSD may show a skewed distribution and very high intra- and inter-individual variation between 10-300 ms. Therefore, a logarithmic representation (e.g., lnRMSSD) may help to increase reproducibility, as well as robustness to artifacts and changes in respiratory patterns. Further, HRV metrics of the frequency domain such as HF or LF may require additional considerations regarding the calculation method (nonparametric vs. parametric, standard spectral analysis or time-frequency analysis), the calculation (absolute or normalized values with or without consideration of very low frequency, VLF), and the duration of measurement. Inconsistent results are often generated using simple ratios, which is therefore discouraged (e.g., the LF / HF ratio may not reliably assess the cardiac sympatho-vagal balance). Insights from non-linear HRV analysis are promising in several clinical and applied settings related to sports medicine and exercise science. As non-linear metrics complement linear HRV and rather focus on (fractal) dynamics and complexity of the time series they might expand our knowledge concerning the behaviour of cardiovascular oscillations in normal healthy conditions as well as in disease states in both resting and exercising conditions. However, these measures still need to be further elucidated for a general practical use.
Context-sensitive analysis and interpretation
Singular, absolute values of HRV (e.g., for RMSSD) provide only limited information; this also applies to the comparison with reference values, which is primarily recommended for specific and homogeneous populations (e.g., diseases, age groups). Comparing a regular intra-individual baseline may help to interpret daily variations in HRV and a rolling average of mean values is favourable for monitoring processes (e.g., 7-day moving average of at least 3 to 5 measurements a week), also in relation to a normal range reference (like the smallest worthwhile change, SWC) for trend analysis of HRV variations. In addition, it may be useful to calculate the coefficient of variation (CV) as a standardized measure of dispersion, calculated by dividing the standard deviation by the mean, in order to gain an impression of the variations; stable regulation can indicate good adaptation to the stress situation (training, everyday life, etc.). You can find two examples below:
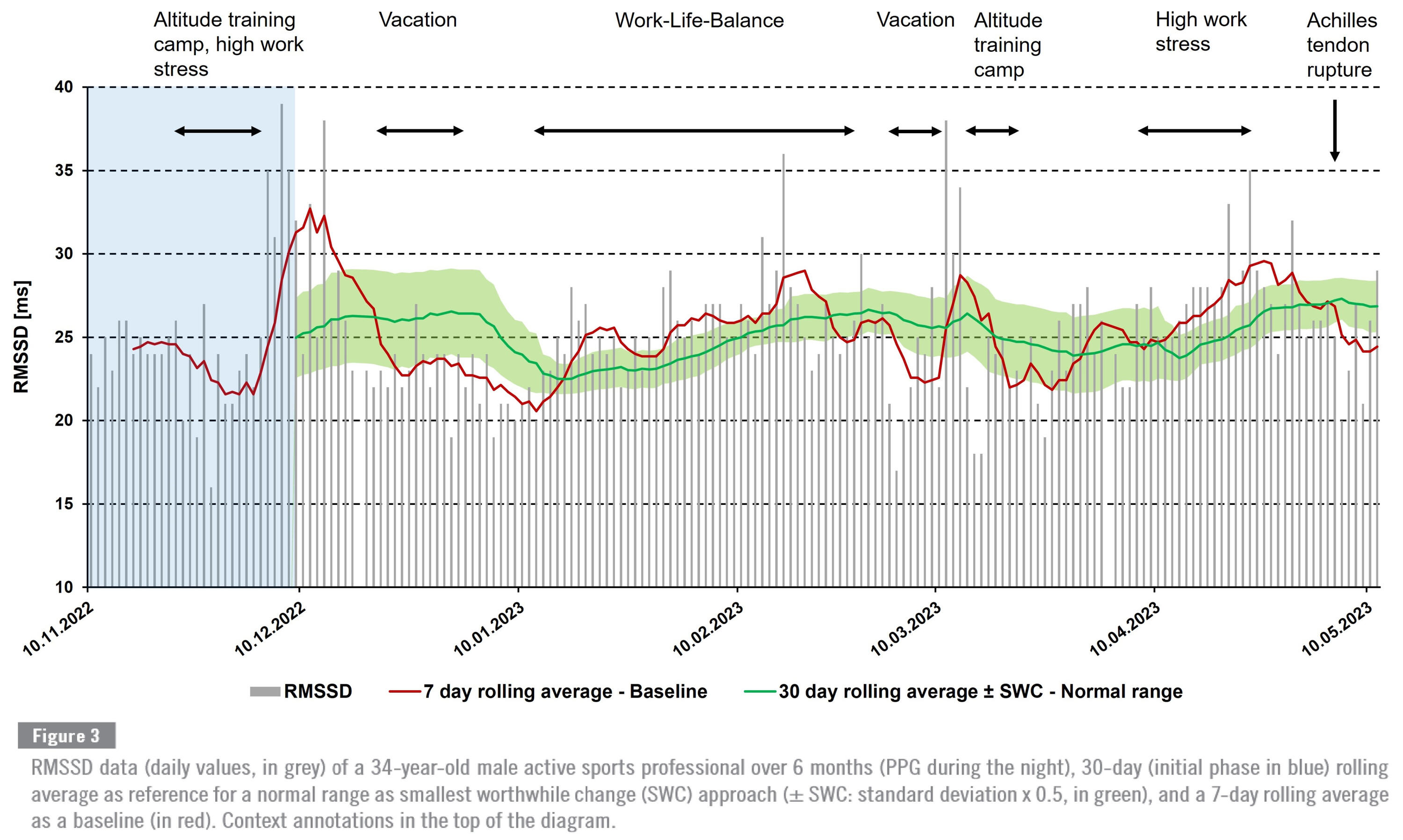
Within the measurement context, it must be decided which wearable or device is used and when an adequate, standardizable measurement context is guaranteed. In the above example, a nighttime measurement was performed using a ring with PPG. It should be noted that the wearable includes the entire night in the analysis, as otherwise the data is dependent on the quality of sleep phase detection. In addition, nighttime analyses may reflect late stressors shortly before bedtime (e.g., late meals, late high-intensity training sessions); this should be taken into account in terms of interpretation. Measurements and analyses in the context of morning measurements immediately after waking up are more likely to be used to assess a situation for the beginning of the day, because the stressors of the previous day may have been processed during sleep. However, not everyone can guarantee a standardized situation early in the morning for regular HRV measurement (e.g., children, pets). A first comparison of nighttime and morning measurements was conducted by Nuuttila and colleagues.
In an overview about the usage of wearable data, including HRV, Marco Altini also displays sound principles to enable practical, meaningful application and interpretation.
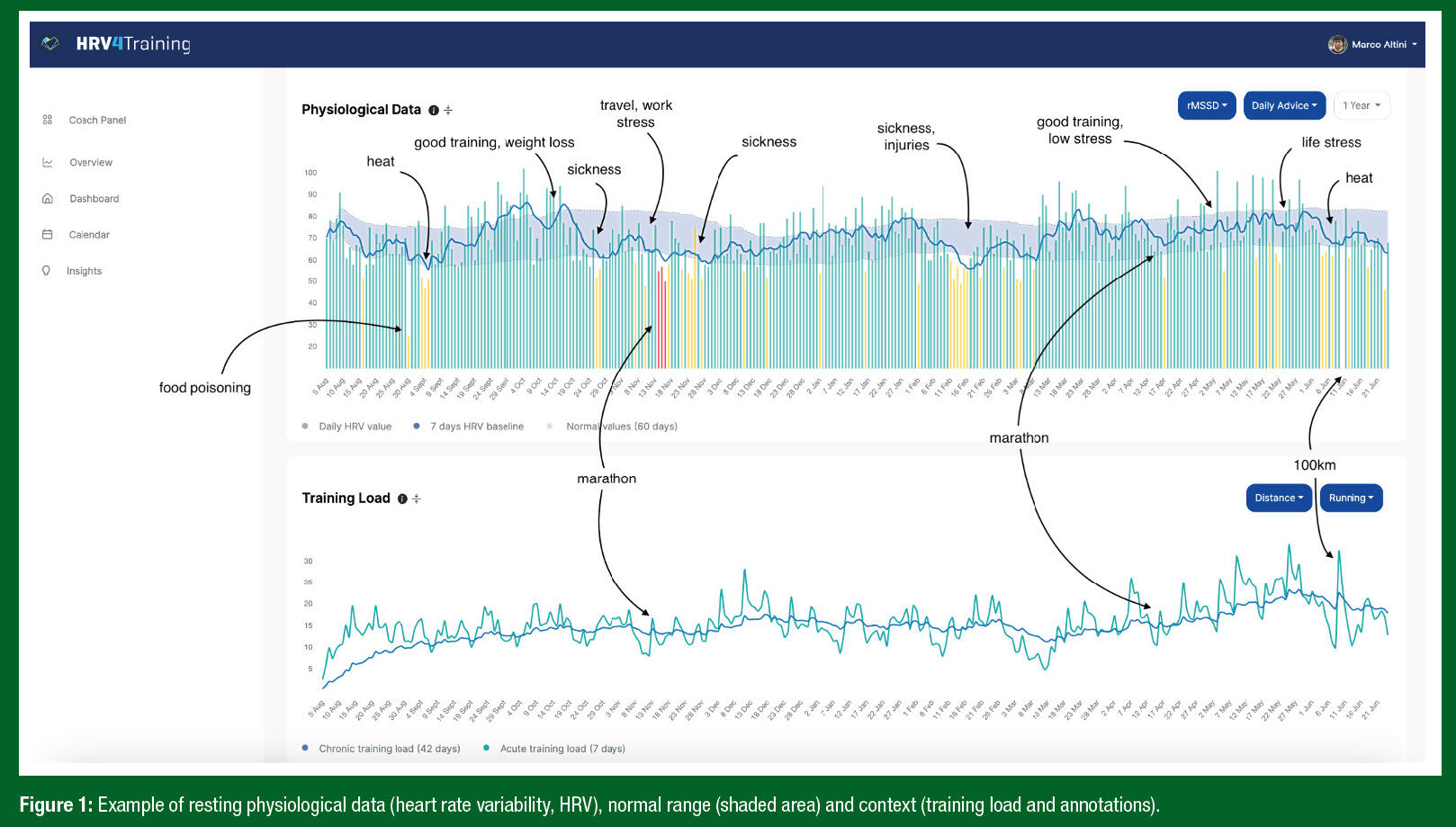

Further information like resting HR, psychometrical scales and other contextual information may overcome the lack of specificity in the interpretation of HRV values. Application of existing standards is mandatory to derive appropriate interpretations. However, higher values (trends) of vagally driven time- and frequency-domain metrics under resting conditions are generally associated with more functionally efficient cardiac autonomic control, whereas HRV declines and becomes more regular with age or disease, revealing a loss of variability and complexity. Lifestyle factors (e.g., physical activity, smoking behavior, alcohol consumption, body composition) are associated with adaptations of cardiac vagal function in this context, so a healthy lifestyle can be considered preventive for reducing age-related decline of positive autonomic regulatory dynamics. The usefulness of HRV as an indicator of physiological and pathological conditions, for risk stratification, and as a marker of autonomic adaptive and regulatory capacity is evident. However, longitudinal data recordings are recommended when trend analysis with contextual data is intended. In addition, the reference setting of resting conditions in supine position can be complemented by active orthostatic tests (supine vs. standing, see below for further information and recommendations), other stress-related conditions like physical exercise, and recovery measurements as these provide additional information on ANS regulation dynamics. However, special care must be taken with both HRV assessment and analysis during stress/exercise and recovery as increased non-stationarity, degraded signal-to-noise ratio and additional physiological phenomena (e.g., cardio-respiratory-locomotor-coupling) may complicate its straightforward use and interpretation.The following study is recommended for the interpretation and context of HRV measurements and the authors conclude:„A camera-based, smartphone app could be used to collect longitudinal data in free-living in a large sample of individuals. Using a simple, one minute measurement upon waking, we could confirm the results of previously published studies as well as provide additional insights on the relationship between resting HR, HRV, population-level characteristics, and acute stressors. There are important implications to these findings. HRV is currently used in sports settings as well as for health and fitness tracking in the general population. However, targeting improvements in HRV as intervention goals might not be realistic, given the strong heritability coupled with age and low explained variance associated with lifestyle factors such as physical activity levels. On the other hand, HRV is able to capture day-to-day stressors within individuals with high sensitivity and might therefore be better suited for such day-to-day stress assessment and management. HRV might be used to infer changes in, e.g., training, while resting HR might be better suited for observing changes in larger stressors, such as sickness. We conclude that resting HR and HRV can effectively be used to quantify individual stress responses across a large range of individual characteristics and stressors. Individual awareness of stress responses might facilitate training guidance, behavioral change, and just-in-time interventions in the future.“
Considering the mentioned context-sensitive requirements, HRV analysis allows for longitudinal trend analysis of patients (including the potential as a remote digital biomarker) and healthy individuals including athletic and non-athletic populations in various clinical and performance-related settings. This includes the application of HRV monitoring for resting conditions, during and/or after biofeedback and training interventions (e.g., breathing interventions, physiological state analysis, intensity domain delineation), as well as general relationships between recovery status, previous exercise conditions, and symptoms of overreaching and overtraining.In regards of longitudinal data analysis, we highlight the advantages of examining temporal dynamics with ambulatory data assessments (e.g., wearables) via micro-longitudinal studies in our new „Perspectives for Progress“ article in „Exercise & Sports Science Reviews“ - using the example of the emerging field of physical activity and cognition. This study approach characterizes frequent within-subject real-life-embedded assessment methods of exposure(s) and outcome(s) in relatively short periods. In addition to the utility to address outstanding research questions, micro-longitudinal studies with ambulatory assessments are also well-situated to complement the evidence obtained from traditional study approaches by strengthening the capacity for causal inferences via a methodological and/or data triangulation, and better informing more resource-intensive, causality-confirming study approaches.In our specific research, we are particularly interested in contextual information necessary to interpret, e.g., psycho-physiological wearable monitoring data (with potential large day-to-day variation and multiple confounding factors such as HRV data), which can be implemented very well within this methodological study approach. Compared to highly controlled intervention studies (e.g., randomized controlled trials), observational studies (e.g., longitudinal studies) provide the key advantages that they are typically (1) less resource-consuming and expensive, (2) have less restrictive exclusion criteria, which strengthens the representability and generalizability with high ecological validity, and (3) less prone to expectancy bias as no intervention is offered.
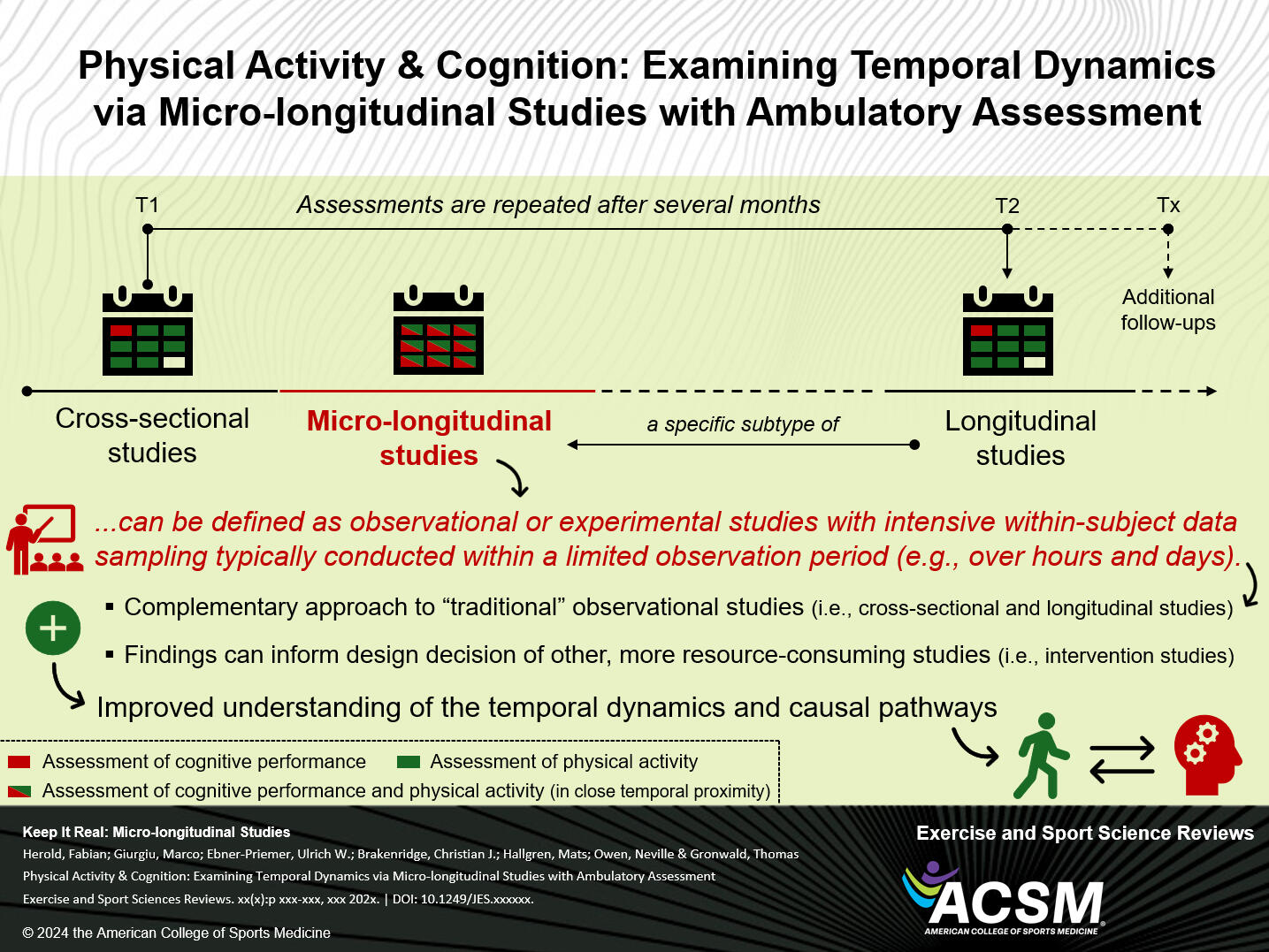
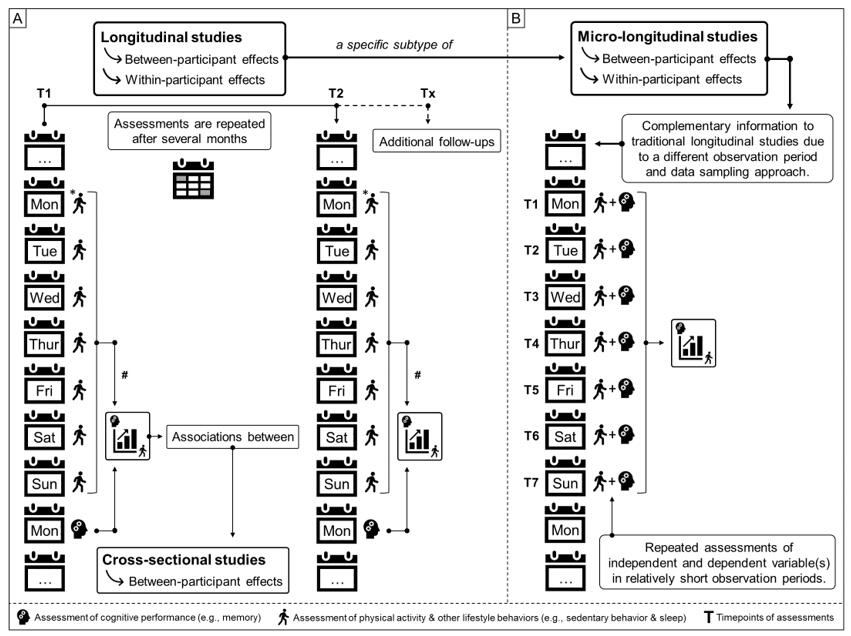
Example studies for monitoring and exercise prescription
In first studies by Kiviniemi and colleagues it could be shown that cardiorespiratory fitness can be effectively improved during endurance training by using daily HRV (from HF domain) for exercise prescription. A lower-intensity training stimulus, whenever attenuated vagally mediated HRV occurs, might be beneficial to gain favorable response in endurance training.
The authors conclude that cardiorespiratory fitness can be effectively improved during endurance training by using daily HRV for exercise prescription and may have benefits over subjectively decided training periodization in moderately active men. Because women are more susceptible to longer recovery of HRV after a vigorous-intensity training, a different HRV-guided training program, where vigorous-intensity training is performed on days with increased HRV, is required for women. By these means, women benefit from HRV guidance by achieving similar improvements in cardiovascular fitness by performing less vigorous-intensity exercises.
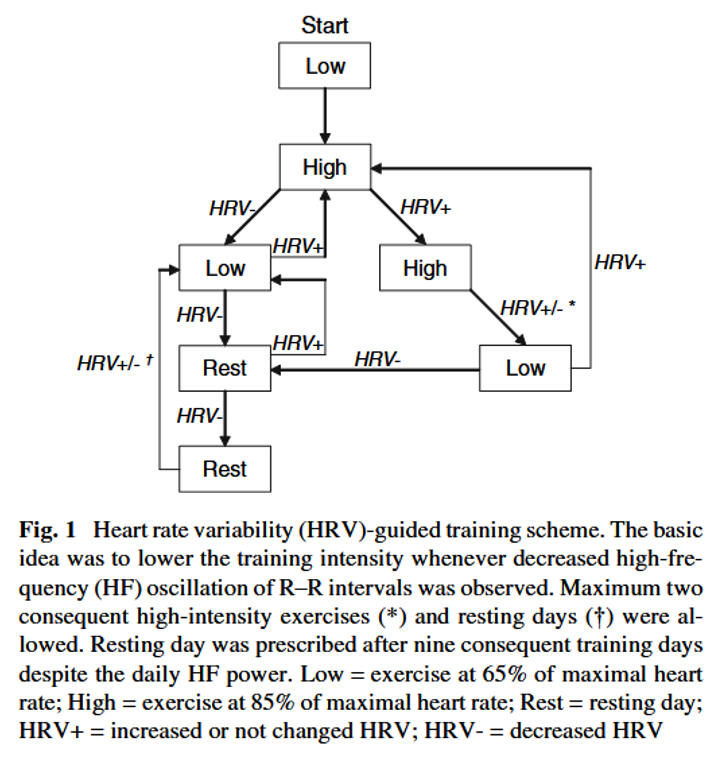
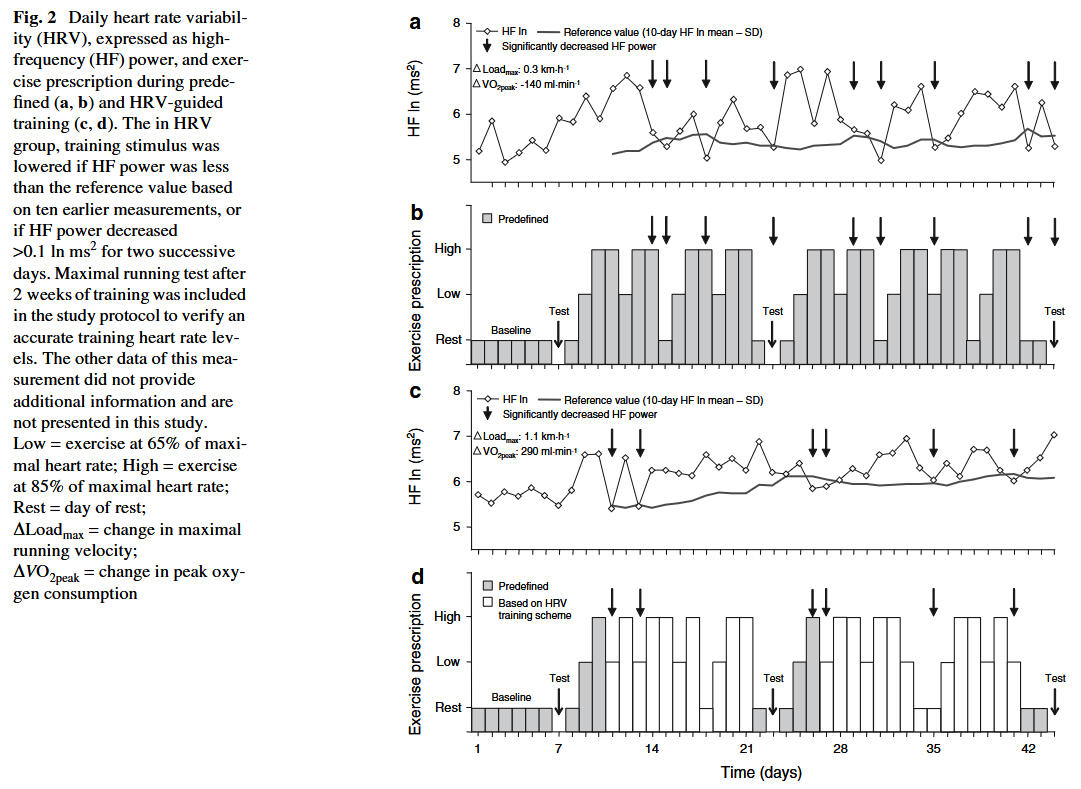
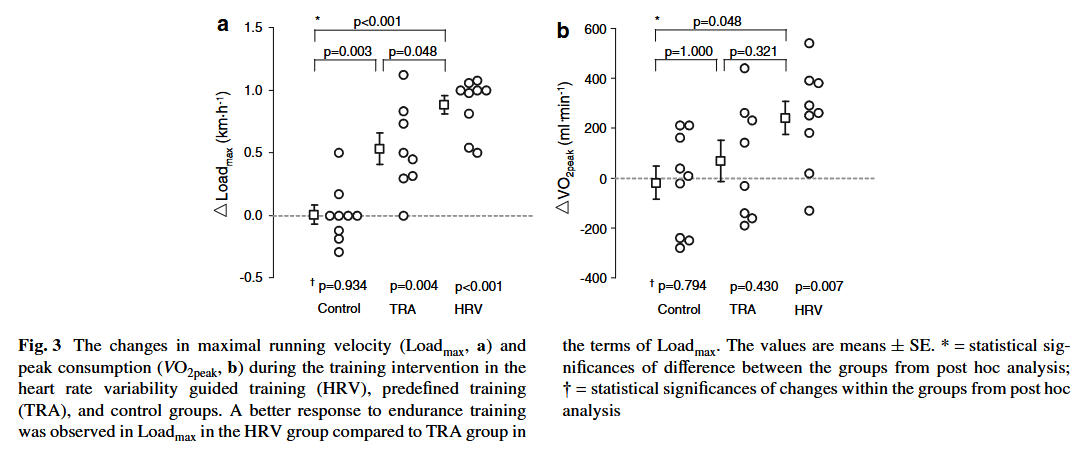
In recent systematic reviews with meta-analysis Düking and colleagues concluded that 4-8 weeks of either HRV-based training or predefined training improves running performance and certain related physiological parameters like submaximal and maximal oxygen consumption. Enhancement of performance and submaximal physiological adaptations appear to be more pronounced with HRV-based training which involves fewer sessions of moderate and/or intense training. In addition, they found that there were fewer non-responders regarding performance with HRV-based training. These conclusions should be considered to be somewhat preliminary, since the available research in this area is both limited and inconsistent.
It remains unclear how training should best be altered in response to a reduction in HRV. For instance, should training be paused completely or high-intensity training replaced with low-intensity sessions? In addition, none of the articles included strategies for recovery, e.g., immersion in cold water, massages or other measures designed to reduce fatigue following endurance exercise. It is possible that the inclusion of such procedures would be more beneficial to performance and/or physiological variables than alterations in training. In addition, factors other than training, including psychological stress related to work or academic studies, exert an impact on HRV and, therefore, the authors cannot be certain what caused the changes in HRV in the interventions described. In this context, characterization of individual stress would be desirable, potentially leading to strategies for reducing this stress and thereby improving recovery and/or training.In that regard, in their latest study, Nuuttila and co-authors evaluated a holistic approach, combining HRV (Polar Vantage V2, wrist-worn device with PPG technology), subjective measures, and also training data to implement changes in training. Another interesting change in this study was that not only load and intensity were reduced when HRV and subjective data were suboptimal, but they were also slightly increased when the data looked good. As a result, the group using this approach improved their running performance to a greater extent. Most importantly, this approach led to zero low-responders, highlighting once again how stressor timing must be accounted for, for training to be effective.
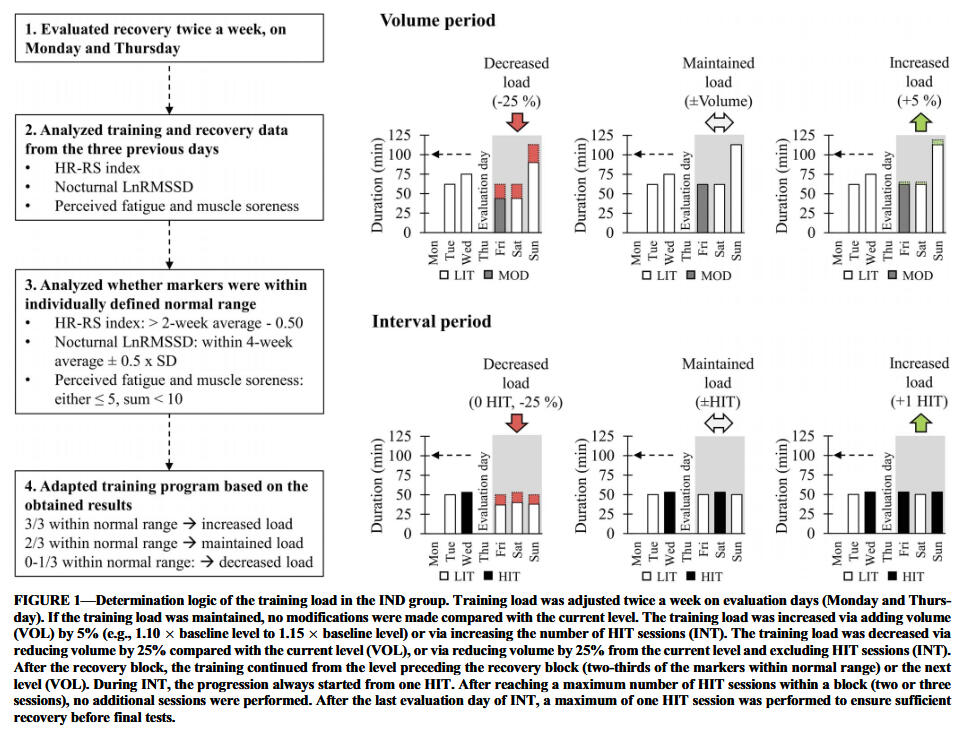
In conclusion, the current study provided evidence that, although predefined training improves endurance performance, individualized endurance training may induce greater improvements in running performance and increase the probability of high response while decreasing the occurrence of low or negative responses to endurance training. In the future, the most suitable markers to be used in monitoring as well as the exact method of how training load could be manipulated during different types of periods should be examined in more detail.Further, in an interesting study from Flatt and colleagues the purpose was to prospectively monitor HRV in college football players throughout an entire competitive season. HRV (RMSSD) was approximated via PPG technology, enabled by an optical pulse-wave finger-sensor and a mobile application from „ithlete“. Time-demand from players was ~3 min/week while waiting to get taped. Though sub-optimal relative to post-waking measures, this approach enabled timely detection of descending patterns, which may be useful for guiding interventions relevant to player health and wellbeing. The authors hypothesized that linemen (predominantly larger players) would experience greater decrements in HRV than nonlinemen.
Tracking HRV in college football players throughout a competitive season revealed progressive autonomic imbalance among the linemen position group. Although significant reductions in LnRMSSD were not observed until the postseason, individual players experienced sustained decrements at earlier time points and in the absence of intensified training. These findings highlight potential deficiencies in or greater taxation to the coping capacity of linemen during the competitive phase. Serial HRV tracking facilitated timely detection of suppressed values, indicating a potential role formobile devices in guiding and monitoring interventions relevant to players’ health and performance.
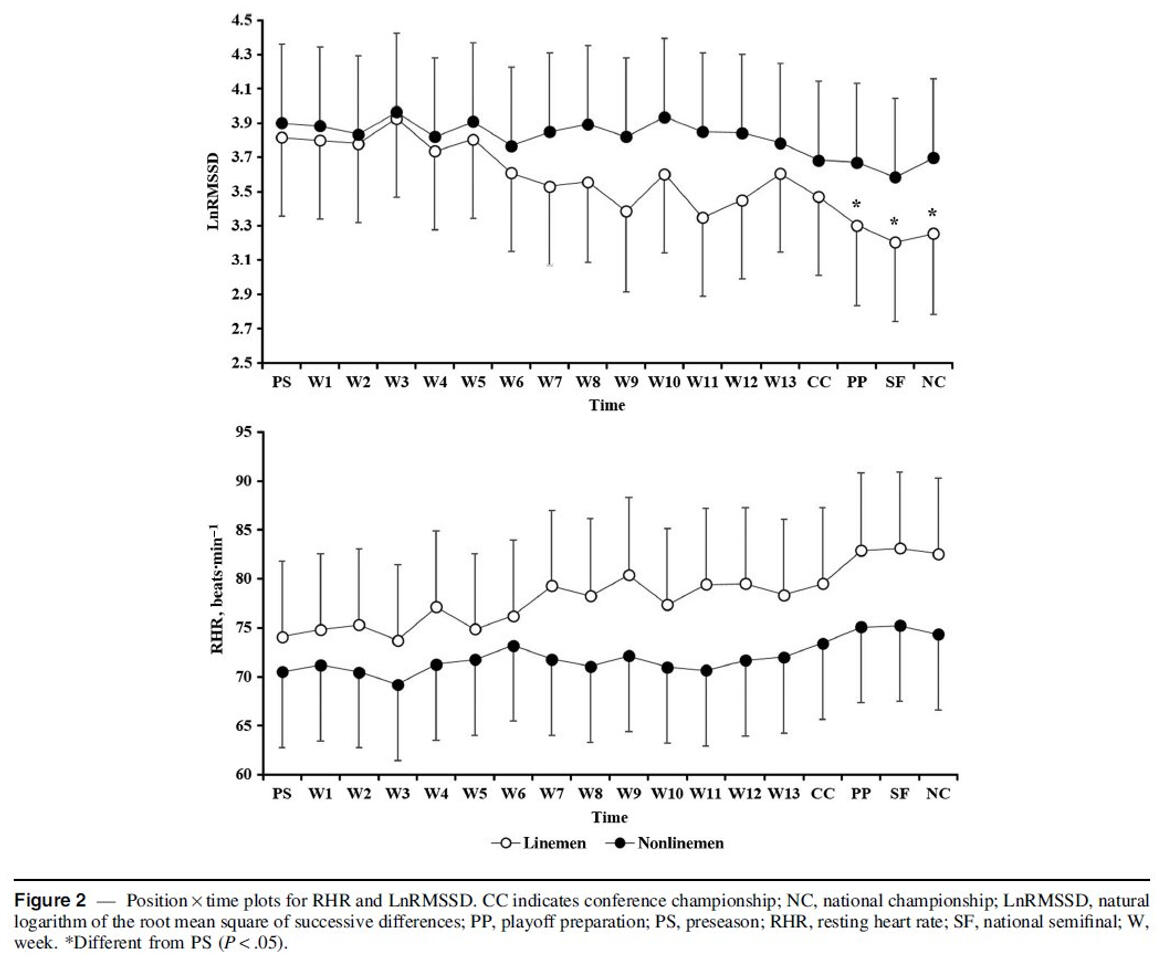
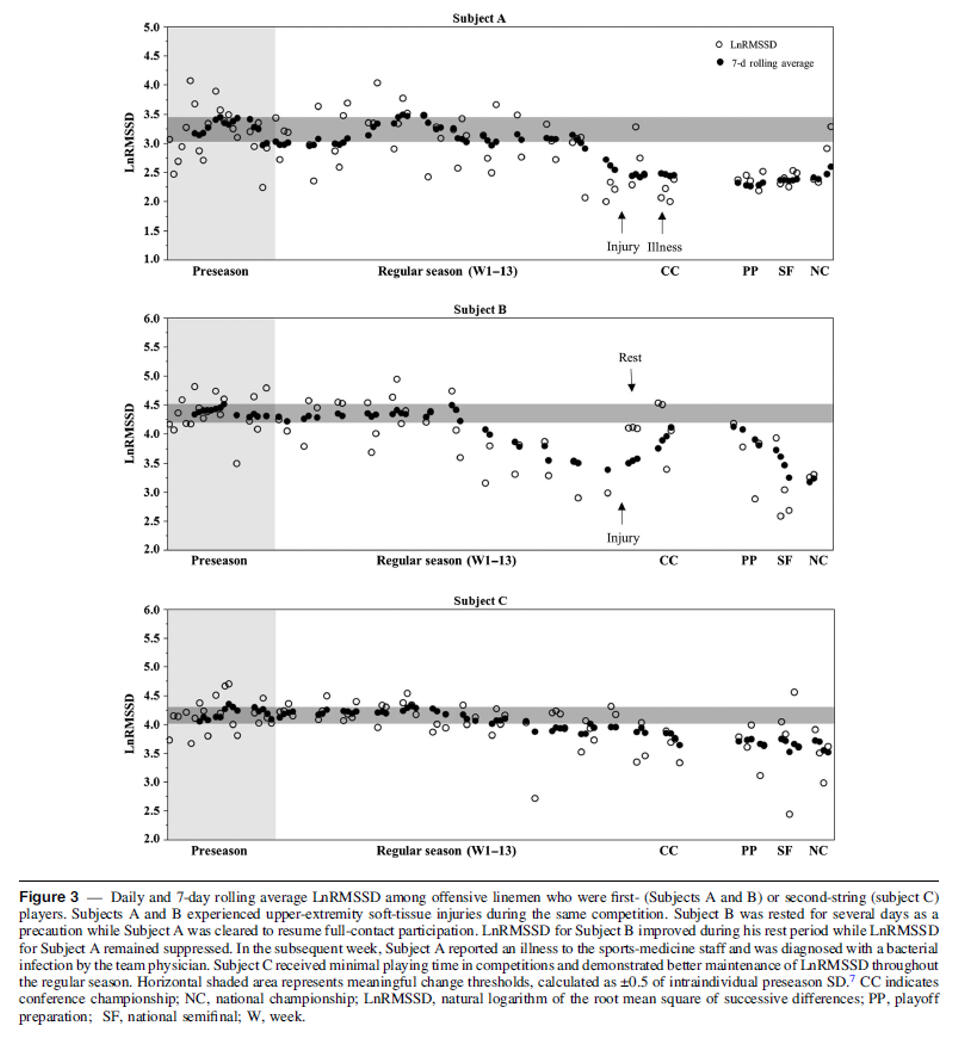
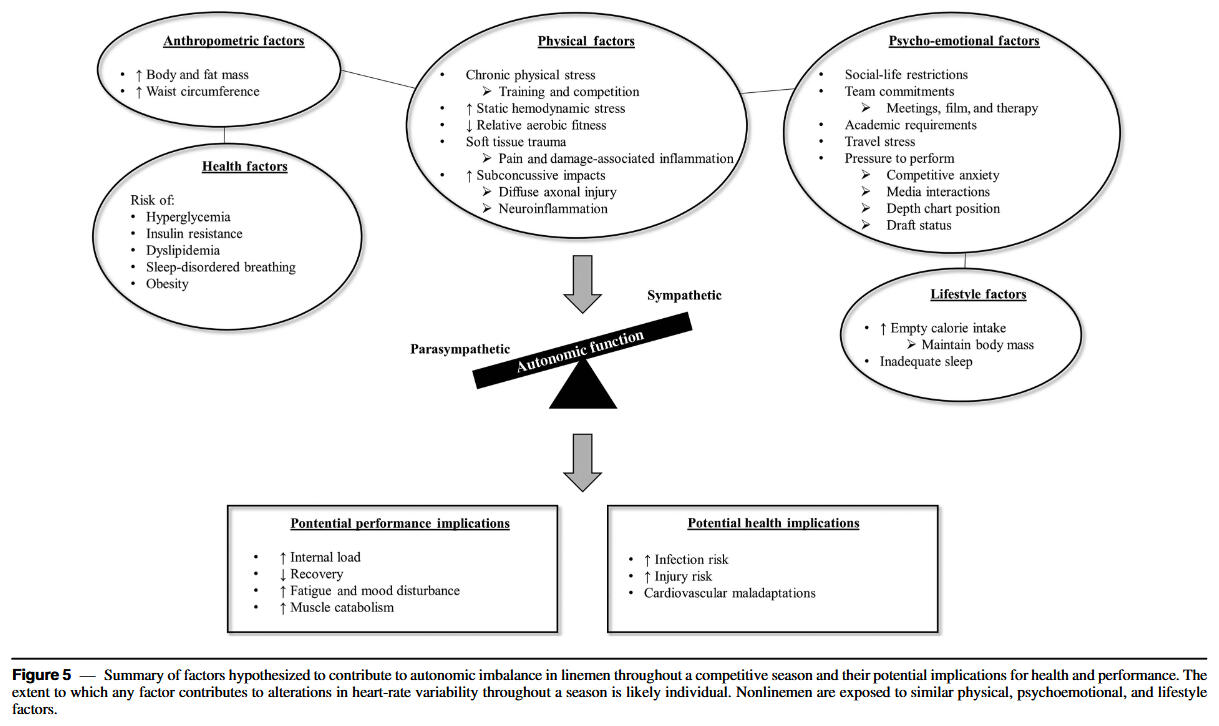
Serial tracking of ultrashort-term HRV with mobile devices at the training facility enabled timely detection of cardiac-parasympathetic suppression among individual players. Factors hypothesized to contribute to autonomic imbalance in linemen and their potential implications for health and performance are summarized in the figure below. Implementation of recovery modalities and lifestyle practices that aim to maintain parasympathetic activation in linemen may limit sustained decrements throughout the season. Due to differences in health conditions, body mass, physical demands, and aerobic fitness among position groups, periodic modification of training frequency or intensity may be required to manage the season-long impact of football participation on autonomic functioning in linemen. Elevations in resting HR conceivably predispose players to concomitant elevations in exercising HR. Thus, such players may experience increased physiological strain (ie, greater internal load) during a typical training session throughout the mid-to-late competitive period.In addition to being a hallmark of impaired recovery, parasympathetic hypoactivity may contribute to cardiovascular maladaptations reported to occur in linemen following a competitive season. Though a better understanding of the health and performance ramifications of suppressed HRV in linemen is needed, a descending pattern in daily values may serve as an easily identifiable red flag requiring attention from performance and medical staff.
The case for heart rate variability reactivity
Laborde et al. (2017) suggest the experimental or practical structure of the three „Rs“ as „Resting“ (baseline), „Reactivity“ (event; stress, physical exercise, change in body position), and „Recovery“ (post-event) measurements. Using this structure allows for the evaluation of tonic HRV for each of this measurement time points and also for the analysis of phasic HRV changes (baseline vs. event like a task, demand: e.g., physical exercise, event vs. post-event, baseline vs. post-event).

In an overview from Manser and colleagues, the authors provide evidence for monitoring phasic responses of HRV in terms of HRV reactivity [i. e., the absolute change from resting state to on-task (i.e., absolute values of HRV measured during exercise)] to provide useful insights into the individual psychophysiological responses of healthy middle-aged to older adults to cognitive and physical exercises.HRV reactivity seems to be a promising biomarker for monitoring internal training load and evaluating neurobiological effects of training interventions. Further research is warranted to evaluate the potential of HRV reactivity as a monitoring parameter to guide cognitive-motor training interventions and/or as a biomarker for cognitive impairment. This may facilitate the early detection of cognitive impairment as well as allow individualized training adaptations that, in turn, support the healthy aging process by optimizing individual exercise dose and progression of cognitive-motor training.
This systematic review with meta-analyses showed that vagally-mediated HRV is significantly reduced during cognitive and/or physical exercises compared to resting state, indicating a significant parasympathetic withdrawal compared to rest. The key moderating variables of these responses identified included exercise intensity for physical exercises, and participant characteristics (i.e., level of cognitive functioning, physical fitness), task demands (i.e., task complexity and modality) and the individual responses to these cognitive challenges for cognitive exercises. In particular, higher task demands (task complexity, physical intensity) were related to larger HRV reactivities. Better physical fitness and cognition were associated with lower HRV reactivities. Additionally, HRV reactivity appeared to be sensitive to training-induced cognitive and neural changes. HRV reactivity seems to be a promising biomarker for monitoring internal training load and evaluating neurobiological effects of training interventions. Further research is warranted to evaluate the potential of HRV reactivity as a monitoring parameter to guide cognitive-motor training interventions and/or as a biomarker for cognitive impairments. This may facilitate the early detection of cognitive impairment as well as allow individualized training adaptations that, in turn, support the healthy aging process by optimizing individual exercise dose and progression of cognitive-motor training.Within this framework, we compiled the potential added value of positional change testing (orthostasis) in the fields of exercise and health science in 2024.
Orthostatic testing, involving the transition from different body positions (e.g., from lying or sitting position to an upright or standing position), offers valuable insights into the autonomic nervous system (ANS) functioning and cardiovascular regulation reflected through complex adjustments in, e.g., measures of HR and HRV. In our narrative review we explore the intricate physiological mechanisms underlying orthostatic stress responses and evaluate its significance for exercise science and sports practice. Into this matter, active orthostatic testing (e.g., active standing up) challenges the cardiovascular autonomic function in a different way than a passive tilt test. It is well documented that there is a transient reduction in blood pressure while standing up, leading to a reflex increase in HR and peripheral vasoconstriction. After that acute response systolic and diastolic blood pressures are usually slightly increased compared to supine lying body position.
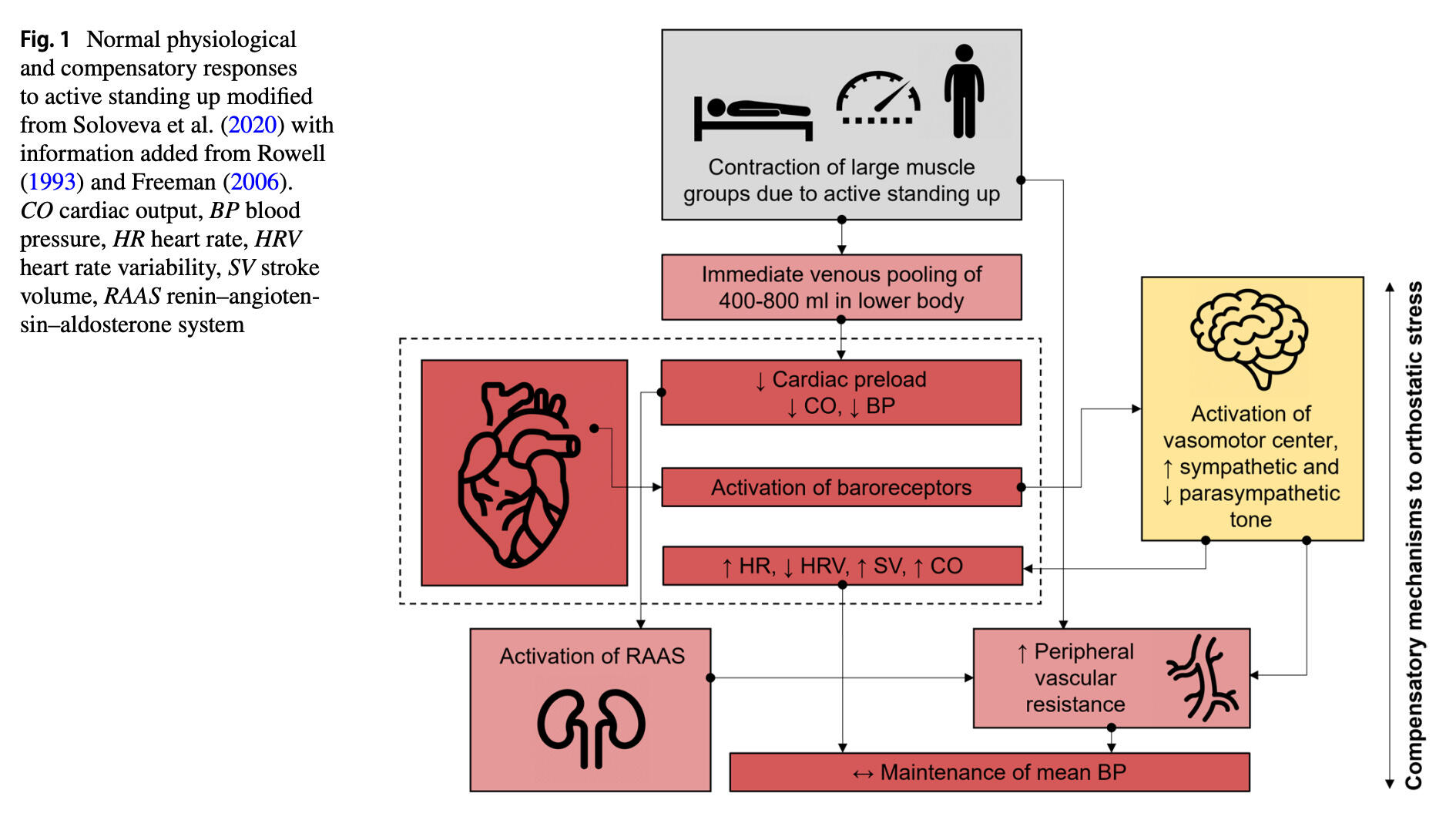
The ANS response to standing is initiated by instantaneous cardiac vagal withdrawal, followed by sympathetic activation and vagal reactivation over the first 25-30 heartbeats. Thus, HR increases immediately upon standing, peaking after 15-20 beats, and is less marked during passive tilting due to the lack of muscular activity. Standing also decreases vagally related HRV indices compared to the supine position.
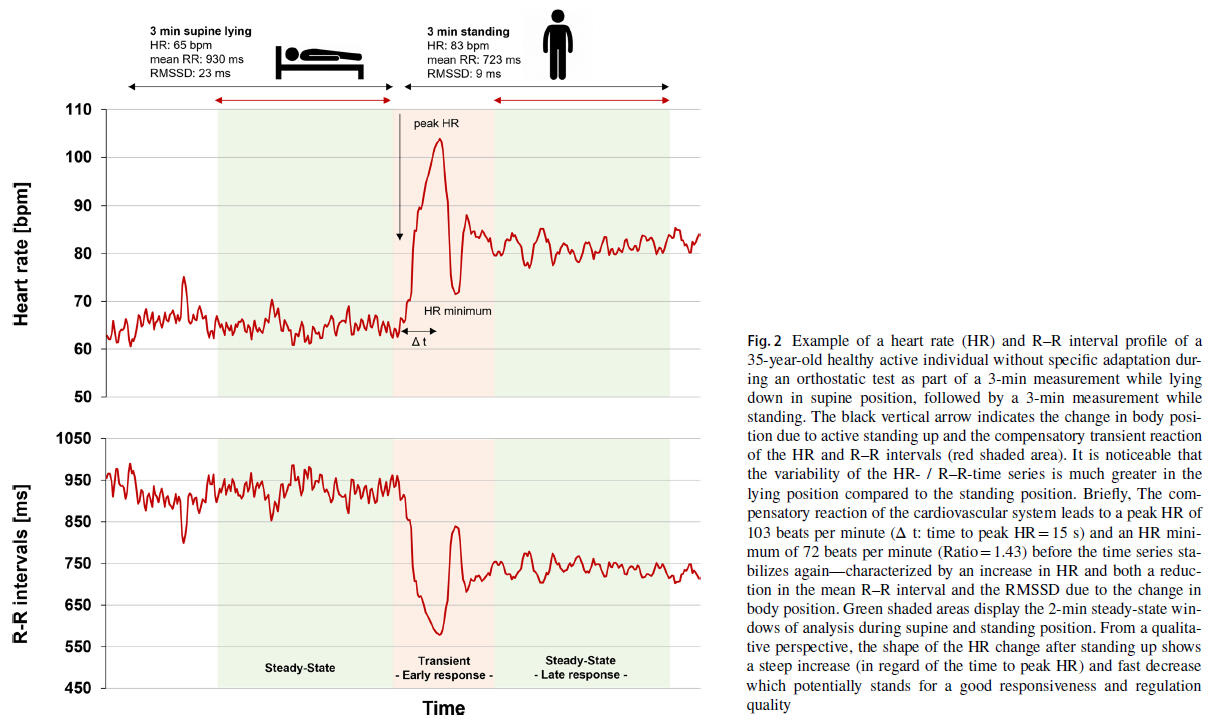
In overtrained endurance athletes, both parasympathetic and sympathetic activity are attenuated in supine and standing positions. Their response to standing is lower than in non-overtrained athletes, with a tendency for further decreased HRV as a sign of pronounced vagal withdrawal and, in some cases, decreased sympathetic excitability, indicating a potential overtraining state. However, as a significant main characteristic, it could be noted that additional pathophysiological conditions consist in a reduced responsiveness or counter-regulation of neural drive in ANS according to an excitatory stimulus, such as an orthostatic challenge. Hence, especially active orthostatic testing could provide additional information about HR(V) reactivity and recovery giving valuable insights into athletes' training status, fatigue levels, and adaptability to workload. Measuring while standing might also counteract the issue of parasympathetic saturation as a common phenomenon especially in well-trained endurance athletes. Data interpretation should be made within intra-individual data history in trend analysis accounting for inter-individual variations in acute responses during testing due to life and physical training stressors. Therefore, additional measures (e.g., psychometrical scales) are required to provide context for HR and HRV analysis interpretation. However, incidence of orthostatic intolerance should be evaluated on an individual level and must be taken into account when considering to implement orthostatic testing in specific subpopulations. Recommendations for standardized testing procedures and interpretation guidelines are developed with the overall aim of enhancing training and recovery strategies. Despite promising study findings in the above-mentioned applied fields, further research, thorough method comparison studies, and systematic reviews are needed to assess the overall perspective of orthostatic testing for training monitoring and fine-tuning of different populations in exercise science and training.If you want to use such testing for your monitoring, take a look at the practical recommendations for implementation at the end of the review; here you will find a list of many potential standardization criteria and possible outcomes for analysis.
Two case studies for monitoring with orthostatic testing
In the following case study, we were able to monitor a viral infection in an elite marathon runner and illustrate the course of the data set before, during, and after infection with relevant outcomes from orthostatic tests.
The findings of this case report have some implications for sports practitioners and coaches looking to both ensure the health of their athletes, and for using HRV as a tool to monitor training process and the return to sport after a viral infection. For endurance athletes, a control by means of resting HR in one body position does not seem to be sufficient. Accordingly, the data have provided supportive rationale as to why the orthostatic test with a change from supine to standing body position and the detection of different indicators based on HR and a vagal driven time-domain HRV parameter (RMSSD) is likely to be useful to detect viral diseases early on when implemented in a daily routine. Given the case study nature of the findings, future research has to be conducted to investigate whether the use of the orthostatic test might be able to offer an innovative, non-invasive, and time-efficient possibility to detect and evaluate the health status of (elite endurance) athletes.
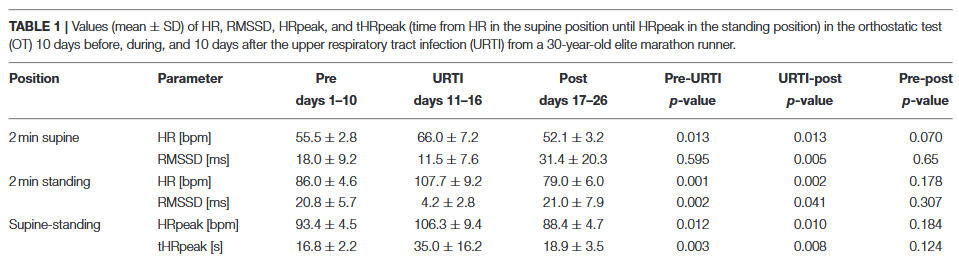
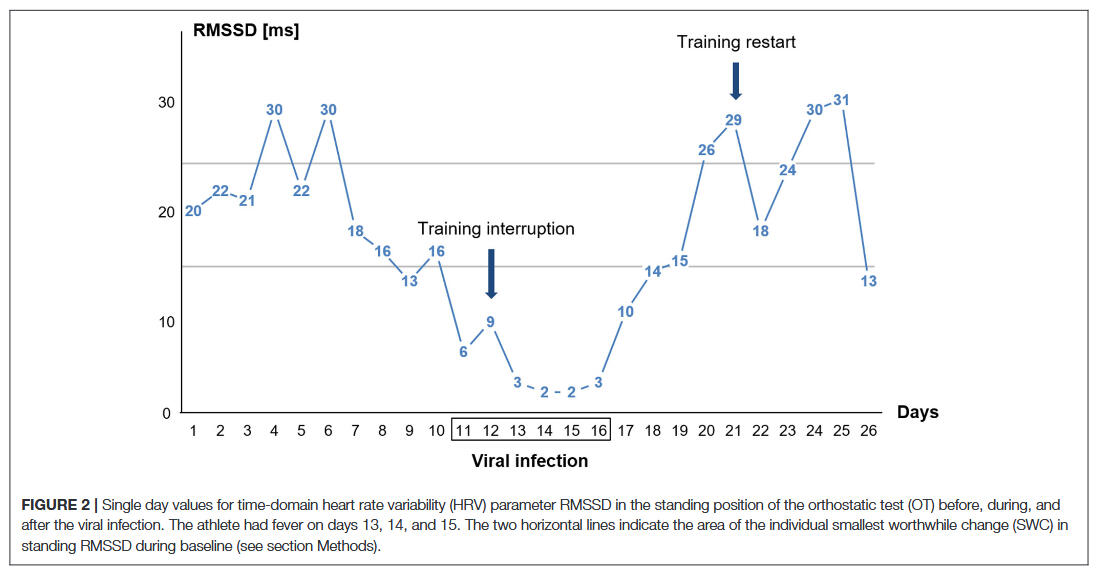
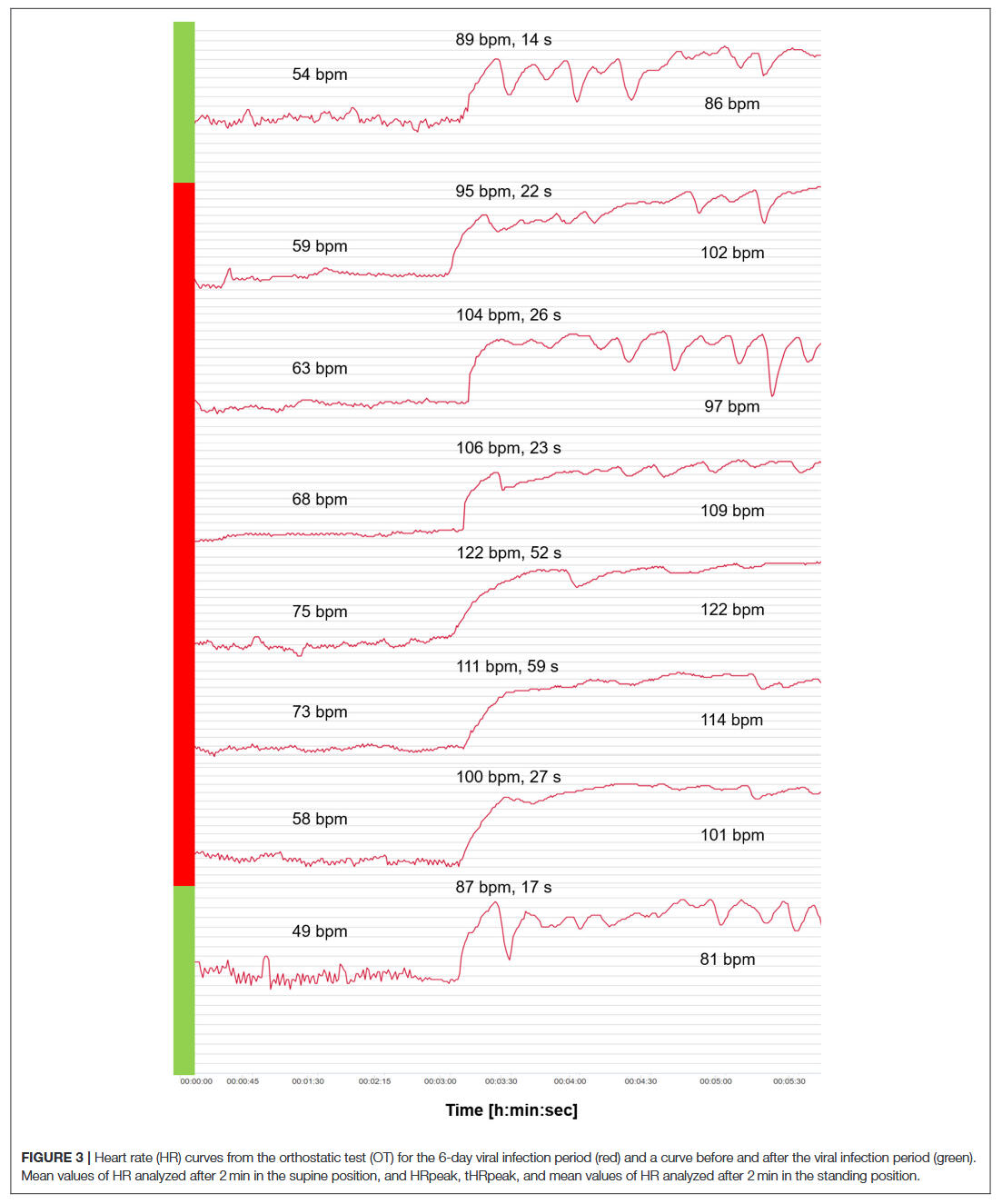
In an early overview from 2013 we could further display a case series of a 22-year-old female middle- and long-distance runner published in an article in German language.
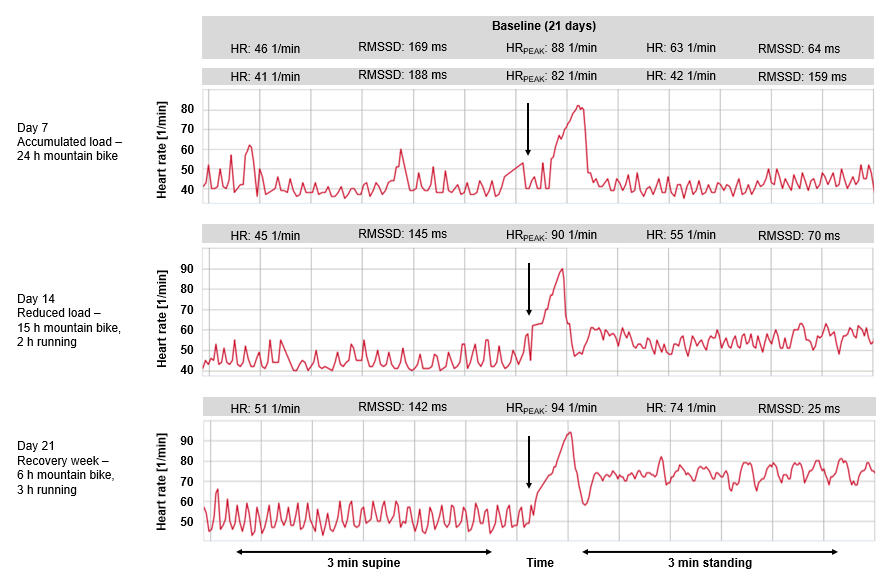
Here, a model is used to illustrate how changes in HR and vagal activity (RMSSD) can be recorded, and then evaluated (the measurement was taken with the Polar V800 and a chest strap with H7 sensor daily for three weeks in the morning after waking up). The values shown are from day 7 after extensive mountain bike (MTB) training (24 hours per week) in the French Alps, from day 14 after 15 hours of MTB and 2 hours of running training with a reduced total volume, and from day 21 after a recovery week with 6 hours of MTB and 3 hours of running training, as well as the average values of the measurements from a baseline over 21 days.The black vertical arrow marks the change in position and the compensatory response of the HR. The autonomic nervous system responds to regular endurance training with pronounced bradycardia as a result of increased vagal tone (HR when lying down or standing of 41 or 42 beats per minute, RMSSD when lying down or standing of 188 or 159 ms). The athlete is not in a state of overtraining, but rather in a functional accumulated load (overreaching) situation, because with a reduction in volume training, vagal tone decreases and the values approach the average values. In order to gain more certainty in the interpretation of the values from the orthostatic tests, additional contextual information such as sleep time, sleep behavior, stress and fatigue level, as well as everyday activities should be analyzed in addition to training.
Practical transfer
Interview for Medscape together with Prof. Cailbhe Doherty
Much has already been said about the application of heart rate variability for monitoring approaches, ... but it can't do any harm to repeat certain important things in a different context; here in an interview for Medscape together with Prof. Calibhe Doherty.The original interview was much longer; so I put it below the image and link.
What is heart rate variability? And is it possible for smartwatches/wearables to accurately measure it?Heart rate (HR) variability (HRV) describes the temporal variation of the heart period or beat-to-beat variability of the HR over a defined measurement period and reflects the dynamic end-organ response of the heart to physiologic and/or pathologic perturbations. This time series of successive time intervals (called as R-R intervals measured in milliseconds; based on the detection of the R-wave in the PQRST complex) is called „tachogram“ from which various metrics with different durations (e.g., from ultra-short-term (1 min), short-term (5min) up to long-term (~24 h) and analysis domains are derived (time-, frequency-, and non-linear domains).In resting conditions it is possible to accurately measure the raw signal of HRV depending on many factors. The gold-standard for a valid and reliable assessment of the time between two successive heartbeats is the electrocardiogram (ECG), which allows recordings in the laboratory or during daily activity (up to 24 hours or longer). In addition, several mobile and user-friendly measurement systems and wearables exist, which can record R-R intervals with different applications and form factors (chest strap systems, adhesive patches and electrode systems on the skin) that also utilize electrophysiological, but also optically derived signals such as photoplethysmography (PPG). The PPG signal can be used to detect HR and HRV also through various form factors such as (wrist) bands, ear or finger clips, rings or even swimming goggles - but more specifically, the signal provides pulse rate (PR) and pulse rate variability (PRV). Most commercially available portable devices show a low absolute error under resting conditions, but should always be validated against reference measures to clarify the accuracy of data parameters and maximize real word application value. It is essential to evaluate the validity depending on the setting, measurement duration, paradigm and cohort that is investigated, because modulators such as the analyzed metric (e.g., root mean square of successive differences of NN intervals, RMSSD), body position, or individual characteristics of the population can cause deviations in HRV measurements from different devices. Therefore, further recommendation is not to compare absolute values between different devices.Is heart rate variability an important measure of someone's health? Is it an important measure for an athlete?HRV and especially relative trends associated with contexual factors can be an important measure for lifestyle and healthcare. Higher values (trends) of vagally driven time- and frequency-domain metrics under resting conditions are generally associated with more functionally efficient cardiac autonomic control, whereas HRV declines and becomes more regular with age or disease, revealing a loss of variability and complexity. Lifestyle factors (e.g., physical activity, smoking behavior, alcohol consumption, body composition) are associated with adaptations of cardiac vagal function in this context, so a healthy lifestyle can be considered preventive for reducing age-related decline of positive autonomic regulatory dynamics. The usefulness of HRV as an indicator of physiological and pathological conditions, for risk stratification, and as a marker of autonomic adaptive and regulatory capacity is evident. However, longitudinal data recordings are recommended when trend analysis with contextual data is intended.Considering the mentioned context-sensitive requirements, HRV analysis allows for longitudinal trend analysis of patients and healthy individuals including athletic and non-athletic populations in various clinical and performance-related settings. This includes the application of HRV monitoring for resting conditions, during and/or after biofeedback and training interventions, as well as general relationships between recovery status, previous exercise conditions, and symptoms of overreaching and overtraining.What should a doctor do if someone goes to them saying their smartwatch is showing that they continuously have a low HRV status? Do you think such data is reliable?This depends strongly on the wearable or recording device. The measurement principles and the type of analysis and processing of the biosignal vary significantly between different devices. On the one hand, sleep analyses are used during the night, while on the other hand, context-free time points during the day are often used. Here, care should be taken to include standardized situations for a measurement and interpretation context. It is recommended to use validated measurement systems that either use the entire night as a reference phase or a consistent measurement situation early in the morning after waking up without prior stressors. Both approaches have advantages and disadvantages. Personally, I cannot use a standardized situation early in the morning because I have two small children, so I have switched to nighttime measurements using a measurement ring; however, these values are highly dependent on late stressors and late influending factors (e.g., late meals or a late training session).In addition, singular, absolute values of HRV (e.g., for RMSSD) provide only limited information; this also applies to the comparison with reference values, which is primarily recommended for specific and homogeneous populations (e.g., diseases, age groups). Comparing a regular intra-individual baseline may help to interpret daily variations in HRV and a rolling average of mean values is favourable for monitoring processes (e.g., 7-day moving average of at least 3 to 5 measurements a week), also in relation to a normal range reference for trend analysis of HRV variations. Further information like resting HR, psychometrical scales and other contextual information may overcome the lack of specificity in the interpretation of HRV values.How can someone improve their HRV?HRV improves when the body is balanced between activity and recovery within homeodynamic boundaries, with strong support from sleep, stress reduction, and a healthy active lifestyle. Regular physical activity, especially moderate aerobic exercise (like walking, cycling, or swimming) and strength training can increase HRV over time. Avoid residual fatigue and overtraining, as excessive exercise may reduce HRV. Breathing exercises with slow, deep, and diaphragmatic breathing (for example, 4–6 breaths per minute) can help to activate the parasympathetic nervous system which improves HRV. Good sleep quality with consistent sleep patterns, enough deep sleep, and avoiding late-night screen time and action are essential for higher HRV. Further, stress management and practices such as meditation, social connection, mindfulness, or progressive muscle relaxation are linked to emotional well-being and can reduce stress and therefore improve HRV. Finally, healthy nutrition habity such as a balanced diet rich in whole foods, omega-3 fatty acids, fruits, and vegetables supports HRV – including the limitation of stimulants, alcohol, nicotine, and processed foods.How important do you think wearable technology is to support the provision of healthcare across Europe? What role do you think it will play in the future?Wearable technology is becoming increasingly important in supporting healthcare across Europe. Devices such as smartwatches, fitness trackers, and medical-grade wearables can provide continuous monitoring of vital signs, heart rhythms (including the detection and analysis of HRV), and physical activity, which helps detect health issues earlier and enables more personalized care. They also empower patients to take a more active role in managing their own health, while giving healthcare providers access to real-time data that can potentially improve decision-making and reduce hospital visits. In the future, wearable technology will likely play a central role in preventive healthcare and chronic disease management, integrating with electronic health records and AI-driven analytics to provide more precise, data-informed treatments. It could also reduce pressure on healthcare systems by shifting some care from hospitals to home environments (also to rural areas without adequate healthcare infrastructure), supporting remote monitoring and telemedicine. Overall, wearables will move from being optional wellness tools to becoming essential components of healthcare delivery across Europe.
HRV explained: Key insights for athletes and coaches - Interview with Ercan Ileri from www.ileriperformance.de
In the video interview in German language we discuss HRV and common questions for application and monitoring purposes for resting physiology assessment:
What is HRV?
What does HRV indicate?
Which factors influence HRV?
How is HRV measured, and what possibilities does it offer for training monitoring and prescription?
What challenges, misconceptions, and limitations exist in the interpretation of HRV data?
What are the key takeaways for athletes and coaches?
„Voll im Takt“ – Our new book in German language
In this book, together with Dr. Alexander Törpel we discuss the broad field of HR measurement in endurance exercise and training. We conclude with a comprehensive overview of monitoring options using HR and HRV.
The hidden flaws of HRV tracking
Everyone’s talking about heart rate variability (HRV) these days, but few realize how easy it is to measure it wrong. Small things like breathing, movement, or even swallowing can throw off your readings. Not to mention all the ways you can misinterpret your HRV readings! In this video, I break down why HRV is more complicated than it seems, why you need a proper protocol, and how to avoid being misled by bad data.
Further links with practical overviews and recommendations
Bibliography
Below you will find a bibliography of reviews and methodological articles, as well as insights about influencing factors and reference values for HRV application; I have also compiled an overview of studies related to practical application and monitoring, including basics and application of orthostatic testing.
Background of heart rate variability under resting, reactivity and recovery conditions:
Acharya, U., Paul Joseph, K., Kannathal, N., Lim, C. M., & Suri, J. S. (2006). Heart rate variability: A review. Medical & Biological Engineering & Computing, 44(12), 1031–1051. https://doi.org/10.1007/s11517-006-0119-0
Achten, J., & Jeukendrup, A. E. (2003). Heart rate monitoring: applications and limitations. Sports Medicine (Auckland, N.Z.), 33(7), 517–538. https://doi.org/10.2165/00007256-200333070-00004
Aubert, A. E., Seps, B., & Beckers, F. (2003). Heart Rate Variability in Athletes: Sports Medicine, 33(12), 889–919. https://doi.org/10.2165/00007256-200333120-00003
Berntson, G. G., Bigger, J. T., Jr, Eckberg, D. L., Grossman, P., Kaufmann, P. G., Malik, M., Nagaraja, H. N., Porges, S. W., Saul, J. P., Stone, P. H., & van der Molen, M. W. (1997). Heart rate variability: origins, methods, and interpretive caveats. Psychophysiology, 34(6), 623–648. https://doi.org/10.1111/j.1469-8986.1997.tb02140.x
Billman, G. E. (2011). Heart Rate Variability? A Historical Perspective. Frontiers in Physiology, 2. https://doi.org/10.3389/fphys.2011.00086
Billman, G. E. (2013). The effect of heart rate on the heart rate variability response to autonomic interventions. Frontiers in Physiology, 4. https://doi.org/10.3389/fphys.2013.00222
Billman, G. E., Huikuri, H. V., Sacha, J., & Trimmel, K. (2015). An introduction to heart rate variability: Methodological considerations and clinical applications. Frontiers in Physiology, 6. https://doi.org/10.3389/fphys.2015.00055
Böckelmann, I. (2012). Analyse der Herzfrequenzvariabilität (HRV)—Praktische Relevanz. Zentralblatt für Arbeitsmedizin, Arbeitsschutz und Ergonomie, 62(5), 275–279. https://doi.org/10.1007/bf03346164
Böckelmann, I., & Sammito, S. (2020). Herzfrequenzvariabilitätsanalyse in der betriebsärztlichen Praxis. Zentralblatt für Arbeitsmedizin, Arbeitsschutz und Ergonomie, 70(6), 269–277. https://doi.org/10.1007/s40664-020-00401-x
Borresen, J., & Lambert, M. I. (2008). Autonomic Control of Heart Rate during and after Exercise: Measurements and Implications for Monitoring Training Status. Sports Medicine, 38(8), 633–646. https://doi.org/10.2165/00007256-200838080-00002
Buchheit, M. (2014). Monitoring training status with HR measures: Do all roads lead to Rome? Frontiers in Physiology, 5, 73. https://doi.org/10.3389/fphys.2014.00073
Cassirame, J., Chevrolat, S., & Mourot, L. (2019). Effects of R-R time series accuracy on heart rate variability indexes. Movement & Sport Sciences - Science & Motricité, 106, 27–35. https://doi.org/10.1051/sm/2019006
Catai, A. M., Pastre, C. M., Godoy, M. F. de, Silva, E. da, Takahashi, A. C. de M., & Vanderlei, L. C. M. (2020). Heart rate variability: Are you using it properly? Standardisation checklist of procedures. Brazilian Journal of Physical Therapy, 24(2), 91–102. https://doi.org/10.1016/j.bjpt.2019.02.006
Ciccone, A. B., Siedlik, J. A., Wecht, J. M., Deckert, J. A., Nguyen, N. D., & Weir, J. P. (2017). Reminder: RMSSD and SD1 are identical heart rate variability metrics. Muscle & Nerve, 56(4), 674–678. https://doi.org/10.1002/mus.25573
Dobbs, W. C., Fedewa, M. V., MacDonald, H. V., Holmes, C. J., Cicone, Z. S., Plews, D. J., & Esco, M. R. (2019). The Accuracy of Acquiring Heart Rate Variability from Portable Devices: A Systematic Review and Meta-Analysis. Sports Medicine, 49(3), 417–435. https://doi.org/10.1007/s40279-019-01061-5
Dong, J.-G. (2016). The role of heart rate variability in sports physiology. Experimental and Therapeutic Medicine, 11(5), 1531–1536. https://doi.org/10.3892/etm.2016.3104
Draghici, A. E., & Taylor, J. A. (2016). The physiological basis and measurement of heart rate variability in humans. Journal of Physiological Anthropology, 35(1), 22. https://doi.org/10.1186/s40101-016-0113-7
Ernst, G. (2017a). Heart-Rate Variability—More than Heart Beats? Frontiers in Public Health, 5, 240. https://doi.org/10.3389/fpubh.2017.00240
Ernst, G. (2017b). Hidden Signals—The History and Methods of Heart Rate Variability. Frontiers in Public Health, 5. https://doi.org/10.3389/fpubh.2017.00265
Grant, C. C., Murray, C., Janse van Rensburg, D. C., & Fletcher, L. (2013). A comparison between heart rate and heart rate variability as indicators of cardiac health and fitness. Frontiers in Physiology, 4, 337. https://doi.org/10.3389/fphys.2013.00337
Grippo, A. J. (2017). Opinion: “Heart Rate Variability, Health and Well-Being: A Systems Perspective” Research Topic. Frontiers in Public Health, 5, 246. https://doi.org/10.3389/fpubh.2017.00246
Gronwald, T., Schaffarczyk, M., Reinsberger, C., & Hoos, O. (2024). Heart Rate Variability – Methods and Analysis in Sports Medicine and Exercise Science. Deutsche Zeitschrift Für Sportmedizin, 75(3), 113–118. https://doi.org/10.5960/dzsm.2024.595
Hoos, O. (2019). Herzratenvariabilität. In M. Frass & L. Krenner (Hrsg.), Integrative Medizin (S. 181–197). Springer Berlin Heidelberg. https://doi.org/10.1007/978-3-662-48879-9_10
Hottenrott, K., Hoos, O., & Esperer, H. D. (2006). Herzfrequenzvariabilität und Sport: Aktueller Stand. Herz, 31(6), 544–552. https://doi.org/10.1007/s00059-006-2855-1
Hottenrott K. & Hoos O. (2017). Heart rate variability analysis in exercise physiology. In: H. Jelinek, A. Khandoker, & D. Cornforth (Eds.), ECG time series analysis: Engineering to medicine (pp. 245–257), London, UK: CRC Press.
Karemaker, J. M. (1997). Heart rate variability: Why do spectral analysis? Heart, 77(2), 99–101. https://doi.org/10.1136/hrt.77.2.99
Karemaker, J. M. (2020). Interpretation of Heart Rate Variability: The Art of Looking Through a Keyhole. Frontiers in Neuroscience, 14, 609570. https://doi.org/10.3389/fnins.2020.609570
Kleiger, R. E., Stein, P. K., & Bigger, J. T. (2005). Heart Rate Variability: Measurement and Clinical Utility. Annals of Noninvasive Electrocardiology, 10(1), 88–101. https://doi.org/10.1111/j.1542-474X.2005.10101.x
Laborde, S., Mosley, E., & Mertgen, A. (2018). A unifying conceptual framework of factors associated to cardiac vagal control. Heliyon, 4(12), e01002. https://doi.org/10.1016/j.heliyon.2018.e01002
Laborde, S., Mosley, E., & Thayer, J. F. (2017). Heart Rate Variability and Cardiac Vagal Tone in Psychophysiological Research – Recommendations for Experiment Planning, Data Analysis, and Data Reporting. Frontiers in Psychology, 8, 213. https://doi.org/10.3389/fpsyg.2017.00213
Laborde, S., Mosley, E., & Mertgen, A. (2018). Vagal Tank Theory: The Three Rs of Cardiac Vagal Control Functioning – Resting, Reactivity, and Recovery. Frontiers in Neuroscience, 12, 458. https://doi.org/10.3389/fnins.2018.00458
Laborde, S., Mosley, E., Bellenger, C., & Thayer, J. (2022). Editorial: Horizon 2030: Innovative Applications of Heart Rate Variability. Frontiers in Neuroscience, 16, 937086. https://doi.org/10.3389/fnins.2022.937086
Malik, M., Hnatkova, K., Huikuri, H. V., Lombardi, F., Schmidt, G., & Zabel, M. (2019). CrossTalk proposal: Heart rate variability is a valid measure of cardiac autonomic responsiveness. The Journal of Physiology, 597(10), 2595–2598. https://doi.org/10.1113/JP277500
Massaro, S., & Pecchia, L. (2019). Heart Rate Variability (HRV) Analysis: A Methodology for Organizational Neuroscience. Organizational Research Methods, 22(1), 354–393. https://doi.org/10.1177/1094428116681072
Mccraty, R., & Shaffer, F. (2015). Heart Rate Variability: New Perspectives on Physiological Mechanisms, Assessment of Self-regulatory Capacity, and Health Risk. Global Advances in Health and Medicine, 4(1), 46–61. https://doi.org/10.7453/gahmj.2014.073
Medeiros, A. R., Michael, S., & Boullosa, D. A. (2018). Make it easier! Evaluation of the ‘vagal-sympathetic effect’ in different conditions with R–R intervals monitoring. European Journal of Applied Physiology, 118(6), 1287–1288. https://doi.org/10.1007/s00421-018-3855-6
Mosley, E., & Laborde, S. (2024). A scoping review of heart rate variability in sport and exercise psychology. International Review of Sport and Exercise Psychology, 17(2), 773–847. https://doi.org/10.1080/1750984X.2022.2092884
Mühlen, J. M., Stang, J., Lykke Skovgaard, E., Judice, P. B., Molina-Garcia, P., Johnston, W., Sardinha, L. B., Ortega, F. B., Caulfield, B., Bloch, W., Cheng, S., Ekelund, U., Brønd, J. C., Grøntved, A., & Schumann, M. (2021). Recommendations for determining the validity of consumer wearable heart rate devices: expert statement and checklist of the INTERLIVE Network. British journal of sports medicine, 55(14), 767–779. https://doi.org/10.1136/bjsports-2020-103148
Owens, A. P. (2020). The Role of Heart Rate Variability in the Future of Remote Digital Biomarkers. Frontiers in Neuroscience, 14, 582145. https://doi.org/10.3389/fnins.2020.582145
Peltola, M. A. (2012). Role of editing of R–R intervals in the analysis of heart rate variability. Frontiers in Physiology, 3. https://doi.org/10.3389/fphys.2012.00148
Quintana, D. S. (2017). Statistical considerations for reporting and planning heart rate variability case‐control studies. Psychophysiology, 54(3), 344–349. https://doi.org/10.1111/psyp.12798
Quintana, D. S., & Heathers, J. A. J. (2014). Considerations in the assessment of heart rate variability in biobehavioral research. Frontiers in Psychology, 5, 805. https://doi.org/10.3389/fpsyg.2014.00805
Quintana, D. S., Alvares, G. A., & Heathers, J. A. J. (2016). Guidelines for Reporting Articles on Psychiatry and Heart rate variability (GRAPH): Recommendations to advance research communication. Translational Psychiatry, 6(5), e803–e803. https://doi.org/10.1038/tp.2016.73
Rincon Soler, A. I., Silva, L. E. V., Fazan, R., & Murta, L. O. (2018). The impact of artifact correction methods of RR series on heart rate variability parameters. Journal of Applied Physiology, 124(3), 646–652. https://doi.org/10.1152/japplphysiol.00927.2016
Sammito, S., & Böckelmann, I. (2015). Analyse der Herzfrequenzvariabilität: Mathematische Basis und praktische Anwendung. Herz, 40(S1), 76–84. https://doi.org/10.1007/s00059-014-4145-7
Sammito, S., & Böckelmann, I. (2016). Möglichkeiten und Einschränkungen der Herzfrequenzmessung und der Analyse der Herzfrequenzvariabilität mittels mobiler Messgeräte: Eine systematische Literaturübersicht. Herzschrittmachertherapie + Elektrophysiologie, 27(1), 38–45. https://doi.org/10.1007/s00399-016-0419-5
Sammito, S., Thielmann, B., Seibt, R., Klussmann, A., Weippert, M., & Böckelmann, I. (2015). Guideline for the application of heart rate and heart rate variability in occupational medicine and occupational science. ASU International, 19(1), 15. https://doi.org/10.17147/ASUI.2015-06-09-03
Sandercock, G. R. H., & Brodie, D. A. (2006). The use of heart rate variability measures to assess autonomic control during exercise. Scandinavian Journal of Medicine & Science in Sports, 16(5), 302–313. https://doi.org/10.1111/j.1600-0838.2006.00556.x
Sassi, R., Cerutti, S., Lombardi, F., Malik, M., Huikuri, H. V., Peng, C. K., Schmidt, G., & Yamamoto, Y. (2015). Advances in heart rate variability signal analysis: joint position statement by the e-Cardiology ESC Working Group and the European Heart Rhythm Association co-endorsed by the Asia Pacific Heart Rhythm Society. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology, 17(9), 1341–1353. https://doi.org/10.1093/europace/euv015
Shaffer, F., & Combatalade, D. C. (2013). Don’t Add or Miss a Beat: A Guide to Cleaner Heart Rate Variability Recordings. Biofeedback, 41(3), 121–130. https://doi.org/10.5298/1081-5937-41.3.04
Shaffer, F., & Ginsberg, J. P. (2017). An Overview of Heart Rate Variability Metrics and Norms. Frontiers in Public Health, 5, 258. https://doi.org/10.3389/fpubh.2017.00258
Shaffer, F., McCraty, R., & Zerr, C. L. (2014). A healthy heart is not a metronome: An integrative review of the heart’s anatomy and heart rate variability. Frontiers in Psychology, 5, 1040. https://doi.org/10.3389/fpsyg.2014.01040
Shaffer, F., Meehan, Z. M., & Zerr, C. L. (2020). A Critical Review of Ultra-Short-Term Heart Rate Variability Norms Research. Frontiers in Neuroscience, 14, 594880. https://doi.org/10.3389/fnins.2020.594880
Singh, N., Moneghetti, K. J., Christle, J. W., Hadley, D., Froelicher, V., & Plews, D. (2018). Heart Rate Variability: An Old Metric with New Meaning in the Era of Using mHealth technologies for Health and Exercise Training Guidance. Part Two: Prognosis and Training. Arrhythmia & electrophysiology review, 7(4), 247–255. https://doi.org/10.15420/aer.2018.30.2
Singh, N., Moneghetti, K. J., Christle, J. W., Hadley, D., Plews, D., & Froelicher, V. (2018). Heart Rate Variability: An Old Metric with New Meaning in the Era of using mHealth Technologies for Health and Exercise Training Guidance. Part One: Physiology and Methods. Arrhythmia & electrophysiology review, 7(3), 193–198. https://doi.org/10.15420/aer.2018.27.2
Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology (1996). Heart rate variability: standards of measurement, physiological interpretation and clinical use. Circulation, 93(5), 1043–1065.
White, D. W., & Raven, P. B. (2014). Autonomic neural control of heart rate during dynamic exercise: Revisited. The Journal of Physiology, 592(12), 2491–2500. https://doi.org/10.1113/jphysiol.2014.271858
Influencing factors and reference values:
Acharya U, R., N, K., Sing, O. W., Ping, L. Y., & Chua, T. (2004). Heart rate analysis in normal subjects of various age groups. BioMedical Engineering OnLine, 3(1). https://doi.org/10.1186/1475-925x-3-24
Altini, M., & Plews, D. (2021). What Is behind Changes in Resting Heart Rate and Heart Rate Variability? A Large-Scale Analysis of Longitudinal Measurements Acquired in Free-Living. Sensors, 21(23), 7932. https://doi.org/10.3390/s21237932
Bauer, A., Camm, A. J., Cerutti, S., Guzik, P., Huikuri, H., Lombardi, F., Malik, M., Peng, C.-K., Porta, A., Sassi, R., Schmidt, G., Schwartz, P. J., Stein, P. K., & Yamamoto, Y. (2017). Reference values of heart rate variability. Heart Rhythm, 14(2), 302–303. https://doi.org/10.1016/j.hrthm.2016.12.015
Brozat, M., Böckelmann, I., & Sammito, S. (2025). Systematic Review on HRV Reference Values. Journal of Cardiovascular Development and Disease, 12(6), 214. https://doi.org/10.3390/jcdd12060214
Corrales, M. M., Torres, B. de la C., Esquivel, A. G., Salazar, M. A. G., & Naranjo Orellana, J. (2012). Normal values of heart rate variability at rest in a young, healthy and active Mexican population. Health, 04(07), 377–385. https://doi.org/10.4236/health.2012.47060
Fatisson, J., Oswald, V., & Lalonde, F. (2016). Influence Diagram of Physiological and Environmental Factors Affecting Heart Rate Variability: An Extended Literature Overview. Heart International, 11(1), heartint.500023. https://doi.org/10.5301/heartint.5000232
Hernández-Vicente, A., Hernando, D., Santos-Lozano, A., Rodríguez-Romo, G., Vicente-Rodríguez, G., Pueyo, E., Bailón, R., & Garatachea, N. (2020). Heart Rate Variability and Exceptional Longevity. Frontiers in Physiology, 11. https://doi.org/10.3389/fphys.2020.566399
Hottenrott, K., Hoos, O., & Esperer, H. D. (2006). Herzfrequenzvariabilität und Sport: Aktueller Stand. Herz Kardiovaskuläre Erkrankungen, 31(6), 544–552. https://doi.org/10.1007/s00059-006-2855-1
Jarczok, M. N., Koenig, J., Wittling, A., Fischer, J. E., & Thayer, J. F. (2019). First Evaluation of an Index of Low Vagally-Mediated Heart Rate Variability as a Marker of Health Risks in Human Adults: Proof of Concept. Journal of Clinical Medicine, 8(11), 1940. https://doi.org/10.3390/jcm8111940
Laborde, S., Mosley, E., & Mertgen, A. (2018). A unifying conceptual framework of factors associated to cardiac vagal control. Heliyon, 4(12), e01002. https://doi.org/10.1016/j.heliyon.2018.e01002
Massin, M., & von Bernuth, G. (1997). Normal Ranges of Heart Rate Variability During Infancy and Childhood. Pediatric Cardiology, 18(4), 297–302. https://doi.org/10.1007/s002469900178
Natarajan, A., Pantelopoulos, A., Emir-Farinas, H., & Natarajan, P. (2020). Heart rate variability with photoplethysmography in 8 million individuals: A cross-sectional study. The Lancet Digital Health, 2(12), e650–e657. https://doi.org/10.1016/s2589-7500(20)30246-6
Nunan, D., Sandercock, G. R. H., & Brodie, D. A. (2010). A Quantitative Systematic Review of Normal Values for Short-Term Heart Rate Variability in Healthy Adults. Pacing and Clinical Electrophysiology, 33(11), 1407–1417. https://doi.org/10.1111/j.1540-8159.2010.02841.x
Nuuttila, O.-P., Seipäjärvi, S., Kyröläinen, H., & Nummela, A. (2022). Reliability and Sensitivity of Nocturnal Heart Rate and Heart-Rate Variability in Monitoring Individual Responses to Training Load. International Journal of Sports Physiology and Performance, 17(8), 1296–1303. https://doi.org/10.1123/ijspp.2022-0145
Sammito, S., & Böckelmann, I. (2016). Factors influencing heart rate variability. International Cardiovascular Forum Journal, 6. https://doi.org/10.17987/icfj.v6i0.242
Sammito, S., & Böckelmann, I. (2016). Reference values for time- and frequency-domain heart rate variability measures. Heart Rhythm, 13(6), 1309–1316. https://doi.org/10.1016/j.hrthm.2016.02.006
Sammito, S., & Böckelmann, I. (2017). New reference values of heart rate variability during ordinary daily activity. Heart Rhythm, 14(2), 304–307. https://doi.org/10.1016/j.hrthm.2016.12.016
Sammito, S., Sammito, W., & Böckelmann, I. (2016). The circadian rhythm of heart rate variability. Biological Rhythm Research, 47(5), 717–730. https://doi.org/10.1080/09291016.2016.1183887
Sammito, S., Thielmann, B., & Böckelmann, I. (2024). Update: Factors influencing heart rate variability–a narrative review. Frontiers in Physiology, 15. https://doi.org/10.3389/fphys.2024.1430458
Sandercock, G. R. H., Bromley, P. D., & Brodie, D. A. (2005). Effects of Exercise on Heart Rate Variability: Inferences from Meta-Analysis. Medicine & Science in Sports & Exercise, 37(3), 433–439. https://doi.org/10.1249/01.mss.0000155388.39002.9d
Schumann, A., & Bär, K.-J. (2022). Autonomic aging – A dataset to quantify changes of cardiovascular autonomic function during healthy aging. Scientific Data, 9(1). https://doi.org/10.1038/s41597-022-01202-y
Shaffer, F., & Meehan, Z. M. (2020). A Practical Guide to Resonance Frequency Assessment for Heart Rate Variability Biofeedback. Frontiers in Neuroscience, 14, 570400. https://doi.org/10.3389/fnins.2020.570400
Soares-Miranda, L., Sattelmair, J., Chaves, P., Duncan, G. E., Siscovick, D. S., Stein, P. K., & Mozaffarian, D. (2014). Physical Activity and Heart Rate Variability in Older Adults: The Cardiovascular Health Study. Circulation, 129(21), 2100–2110. https://doi.org/10.1161/circulationaha.113.005361
Tegegne, B. S., Man, T., van Roon, A. M., Riese, H., & Snieder, H. (2018). Determinants of heart rate variability in the general population: The Lifelines Cohort Study. Heart Rhythm, 15(10), 1552–1558. https://doi.org/10.1016/j.hrthm.2018.05.006
Tegegne, B. S., Man, T., van Roon, A. M., Snieder, H., & Riese, H. (2020). Reference values of heart rate variability from 10-second resting electrocardiograms: The Lifelines Cohort Study. European Journal of Preventive Cardiology, 27(19), 2191–2194. https://doi.org/10.1177/2047487319872567
Thayer, J. F., Mather, M., & Koenig, J. (2021). Stress and aging: A neurovisceral integration perspective. Psychophysiology, 58(7). https://doi.org/10.1111/psyp.13804
Tornberg, J., Ikäheimo, T. M., Kiviniemi, A., Pyky, R., Hautala, A., Mäntysaari, M., Jämsä, T., & Korpelainen, R. (2019). Physical activity is associated with cardiac autonomic function in adolescent men. PLOS ONE, 14(9), e0222121. https://doi.org/10.1371/journal.pone.0222121
Umetani, K., Singer, D. H., McCraty, R., & Atkinson, M. (1998). Twenty-Four Hour Time Domain Heart Rate Variability and Heart Rate: Relations to Age and Gender Over Nine Decades. Journal of the American College of Cardiology, 31(3), 593–601. https://doi.org/10.1016/s0735-1097(97)00554-8
Uusitalo, A. L. T., Vanninen, E., Levälahti, E., Battié, M. C., Videman, T., & Kaprio, J. (2007). Role of genetic and environmental influences on heart rate variability in middle-aged men. American Journal of Physiology-Heart and Circulatory Physiology, 293(2), H1013–H1022. https://doi.org/10.1152/ajpheart.00475.2006
Voss, A., Schroeder, R., Heitmann, A., Peters, A., & Perz, S. (2015). Short-Term Heart Rate Variability—Influence of Gender and Age in Healthy Subjects. PLOS ONE, 10(3), e0118308. https://doi.org/10.1371/journal.pone.0118308
Practical application and monitoring:
Altini, M. (2024). Making sense of wearables data. Sports Science Exchange, 37(250), 1–6.
Aubert, A. E., Seps, B., & Beckers, F. (2003). Heart Rate Variability in Athletes. Sports Medicine, 33(12), 889–919. https://doi.org/10.2165/00007256-200333120-00003
Bellenger, C. R., Fuller, J. T., Thomson, R. L., Davison, K., Robertson, E. Y., & Buckley, J. D. (2016). Monitoring Athletic Training Status Through Autonomic Heart Rate Regulation: A Systematic Review and Meta-Analysis. Sports Medicine, 46(10), 1461–1486. https://doi.org/10.1007/s40279-016-0484-2
Bellenger, C. R., Karavirta, L., Thomson, R. L., Robertson, E. Y., Davison, K., & Buckley, J. D. (2016). Contextualizing Parasympathetic Hyperactivity in Functionally Overreached Athletes With Perceptions of Training Tolerance. International Journal of Sports Physiology and Performance, 11(5), 685–692. https://doi.org/10.1123/ijspp.2015-0495
Bellenger, C. R., Thomson, R. L., Davison, K., Robertson, E. Y., & Buckley, J. D. (2021). The Impact of Functional Overreaching on Post-exercise Parasympathetic Reactivation in Runners. Frontiers in Physiology, 11. https://doi.org/10.3389/fphys.2020.614765
Bellenger, C. R., Thomson, R. L., Robertson, E. Y., Davison, K., Nelson, M. J., Karavirta, L., & Buckley, J. D. (2017). The effect of functional overreaching on parameters of autonomic heart rate regulation. European Journal of Applied Physiology, 117(3), 541–550. https://doi.org/10.1007/s00421-017-3549-5
Borresen, J., & Lambert, M. I. (2008). Autonomic Control of Heart Rate during and after Exercise: Measurements and Implications for Monitoring Training Status. Sports Medicine, 38(8), 633–646. https://doi.org/10.2165/00007256-200838080-00002
Bosquet, L., Merkari, S., Arvisais, D., & Aubert, A. E. (2008). Is heart rate a convenient tool to monitor over-reaching? A systematic review of the literature. British Journal of Sports Medicine, 42(9), 709–714. https://doi.org/10.1136/bjsm.2007.042200
Bourdillon, N., Bellenoue, S., Schmitt, L., & Millet, G. P. (2024). Daily cardiac autonomic responses during the Tour de France in a male professional cyclist. Frontiers in Neuroscience, 17. https://doi.org/10.3389/fnins.2023.1221957
Buchheit, M. (2014). Monitoring training status with HR measures: Do all roads lead to Rome? Frontiers in Physiology, 5. https://doi.org/10.3389/fphys.2014.00073
Buchheit, M., Chivot, A., Parouty, J., Mercier, D., Al Haddad, H., Laursen, P. B., & Ahmaidi, S. (2010). Monitoring endurance running performance using cardiac parasympathetic function. European Journal of Applied Physiology, 108(6), 1153–1167. https://doi.org/10.1007/s00421-009-1317-x
Buchheit, M., Papelier, Y., Laursen, P. B., & Ahmaidi, S. (2007). Noninvasive assessment of cardiac parasympathetic function: Postexercise heart rate recovery or heart rate variability? American Journal of Physiology-Heart and Circulatory Physiology, 293(1), H8–H10. https://doi.org/10.1152/ajpheart.00335.2007
Buchheit, M., & Gindre, C. (2006). Cardiac parasympathetic regulation: Respective associations with cardiorespiratory fitness and training load. American Journal of Physiology-Heart and Circulatory Physiology, 291(1), H451–H458. https://doi.org/10.1152/ajpheart.00008.2006
Buchheit, M., Simpson, M. B., Al Haddad, H., Bourdon, P. C., & Mendez-Villanueva, A. (2012). Monitoring changes in physical performance with heart rate measures in young soccer players. European Journal of Applied Physiology, 112(2), 711–723. https://doi.org/10.1007/s00421-011-2014-0
Carrasco-Poyatos, M., González-Quílez, A., Altini, M., & Granero-Gallegos, A. (2022). Heart rate variability-guided training in professional runners: Effects on performance and vagal modulation. Physiology & Behavior, 244, 113654. https://doi.org/10.1016/j.physbeh.2021.113654
Carrasco-Poyatos, M., González-Quílez, A., Martínez-González-Moro, I., & Granero-Gallegos, A. (2020). HRV-Guided Training for Professional Endurance Athletes: A Protocol for a Cluster-Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 17(15), 5465. https://doi.org/10.3390/ijerph17155465
Casanova-Lizón, A., Manresa-Rocamora, A., Sarabia, J. M., Pastor, D., Javaloyes, A., Peña-González, I., & Moya-Ramón, M. (2025). Impact of heart rate variability-based exercise prescription: Self-guided by technology and trainer-guided exercise in sedentary adults. Frontiers in Sports and Active Living, 7. https://doi.org/10.3389/fspor.2025.1578478
Doherty, C., Baldwin, M., Lambe, R., Burke, D., & Altini, M. (2025). Readiness, recovery, and strain: An evaluation of composite health scores in consumer wearables. Translational Exercise Biomedicine. https://doi.org/10.1515/teb-2025-0001
Düking, P., Zinner, C., Reed, J. L., Holmberg, H., & Sperlich, B. (2020). Predefined vs data‐guided training prescription based on autonomic nervous system variation: A systematic review. Scandinavian Journal of Medicine & Science in Sports, 30(12), 2291–2304. https://doi.org/10.1111/sms.13802
Düking, P., Zinner, C., Trabelsi, K., Reed, J. L., Holmberg, H.-C., Kunz, P., & Sperlich, B. (2021). Monitoring and adapting endurance training on the basis of heart rate variability monitored by wearable technologies: A systematic review with meta-analysis. Journal of Science and Medicine in Sport, 24(11), 1180–1192. https://doi.org/10.1016/j.jsams.2021.04.012
Dupuit, M., Barlier, K., Tranchard, B., Toussaint, J.-F., Antero, J., & Pla, R. (2025). Heart Rate Variability Measurements Across the Menstrual Cycle and Oral Contraceptive Phases in Two Olympian Female Swimmers: A Case Report. Sports, 13(6), 185. https://doi.org/10.3390/sports13060185
Fazackerley, L. A., Fell, J. W., & Kitic, C. M. (2019). The effect of an ultra-endurance running race on heart rate variability. European Journal of Applied Physiology, 119(9), 2001–2009. https://doi.org/10.1007/s00421-019-04187-6
Figueiredo, D. H., Figueiredo, D. H., Bellenger, C., & Machado, F. A. (2022). Individually guided training prescription by heart rate variability and self-reported measure of stress tolerance in recreational runners: Effects on endurance performance. Journal of Sports Sciences, 40(24), 2732–2740. https://doi.org/10.1080/02640414.2023.2191082
Flatt, A. A., & Howells, D. (2019). Effects of varying training load on heart rate variability and running performance among an Olympic rugby sevens team. Journal of Science and Medicine in Sport, 22(2), 222–226. https://doi.org/10.1016/j.jsams.2018.07.014
Flatt, A. A., Allen, J. R., Keith, C. M., Martinez, M. W., & Esco, M. R. (2021). Season-Long Heart-Rate Variability Tracking Reveals Autonomic Imbalance in American College Football Players. International Journal of Sports Physiology and Performance, 16(12), 1834–1843. https://doi.org/10.1123/ijspp.2020-0801
Flatt, A. A., Esco, M. R., Nakamura, F. Y., & Plews, D. J. (2017). Interpreting daily heart rate variability changes in collegiate female soccer players. The Journal of Sports Medicine and Physical Fitness, 57(6). https://doi.org/10.23736/s0022-4707.16.06322-2
Flatt, A. A., Hornikel, B., & Esco, M. R. (2017). Heart rate variability and psychometric responses to overload and tapering in collegiate sprint-swimmers. Journal of Science and Medicine in Sport, 20(6), 606–610. https://doi.org/10.1016/j.jsams.2016.10.017
Grainger, A., Heffernan, S., Waldron, M., & Sawczuk, T. (2022). Autonomic Nervous System Indices of Player Readiness During Elite-Level Rugby Union Game-Week Microcycles. Journal of strength and conditioning research, 36(11), 3173–3178. https://doi.org/10.1519/JSC.0000000000004292
Granero-Gallegos, A., González-Quílez, A., Plews, D., & Carrasco-Poyatos, M. (2020). HRV-Based Training for Improving VO2max in Endurance Athletes. A Systematic Review with Meta-Analysis. International Journal of Environmental Research and Public Health, 17(21), 7999. https://doi.org/10.3390/ijerph17217999
Gronwald, T., Schaffarczyk, M., & Hoos, O. (2024). Orthostatic testing for heart rate and heart rate variability monitoring in exercise science and practice. European Journal of Applied Physiology, 124(12), 3495–3510. https://doi.org/10.1007/s00421-024-05601-4
Gronwald, T., Schaffarczyk, M., Reinsberger, C., & Hoos, O. (2024). Heart Rate Variability – Methods and Analysis in Sports Medicine and Exercise Science. Deutsche Zeitschrift Für Sportmedizin, 75(3), 113–118. https://doi.org/10.5960/dzsm.2024.595
Guthrie, B., Jagim, A. R., & Jones, M. T. (2023). Ready or Not, Here I Come: A Scoping Review of Methods Used to Assess Player Readiness Via Indicators of Neuromuscular Function in Football Code Athletes. Strength & Conditioning Journal, 45(1), 93–110. https://doi.org/10.1519/ssc.0000000000000735
Hottenrott, K. & Gronwald, T. (2014). Bedeutung der Herzfrequenzvariabilität für die Regenerationssteuerung. Leistungssport, 44(5), 9-13.
Hottenrott, L., Gronwald, T., Hottenrott, K., Wiewelhove, T., & Ferrauti, A. (2021). Utilizing Heart Rate Variability for Coaching Athletes During and After Viral Infection: A Case Report in an Elite Endurance Athlete. Frontiers in Sports and Active Living, 3, 612782. https://doi.org/10.3389/fspor.2021.612782
Hynynen, E., Uusitalo, A., Konttinen, N., & Rusko, H. (2006). Heart Rate Variability during Night Sleep and after Awakening in Overtrained Athletes. Medicine & Science in Sports & Exercise, 38(2), 313–317. https://doi.org/10.1249/01.mss.0000184631.27641.b5
Hynynen, E., Vesterinen, V., Rusko, H., & Nummela, A. (2010). Effects of Moderate and Heavy Endurance Exercise on Nocturnal HRV. International Journal of Sports Medicine, 31(06), 428–432. https://doi.org/10.1055/s-0030-1249625
Javaloyes, A., Mateo-March, M., Manresa-Rocamora, A., Sanz-Quinto, S., & Moya-Ramón, M. (2021). The Use of a Smartphone Application in Monitoring HRV during an Altitude Training Camp in Professional Female Cyclists: A Preliminary Study. Sensors, 21(16), 5497. https://doi.org/10.3390/s21165497
Javaloyes, A., Sarabia, J. M., Lamberts, R. P., & Moya-Ramon, M. (2019). Training Prescription Guided by Heart-Rate Variability in Cycling. International Journal of Sports Physiology and Performance, 14(1), 23–32. https://doi.org/10.1123/ijspp.2018-0122
Katona, P. G., McLean, M., Dighton, D. H., & Guz, A. (1982). Sympathetic and parasympathetic cardiac control in athletes and nonathletes at rest. Journal of Applied Physiology, 52(6), 1652–1657. https://doi.org/10.1152/jappl.1982.52.6.1652
Kiviniemi, A. M., Hautala, A. J., Kinnunen, H., & Tulppo, M. P. (2007). Endurance training guided individually by daily heart rate variability measurements. European Journal of Applied Physiology, 101(6), 743–751. https://doi.org/10.1007/s00421-007-0552-2
Kiviniemi, A. M., Hautala, A. J., Kinnunen, H., Nissilä, J., Virtanen, P., Karjalainen, J., & Tulppo, M. P. (2010). Daily Exercise Prescription on the Basis of HR Variability among Men and Women. Medicine & Science in Sports & Exercise, 42(7), 1355–1363. https://doi.org/10.1249/mss.0b013e3181cd5f39
Lamberts, R. P., Rietjens, G. J., Tijdink, H. H., Noakes, T. D., & Lambert, M. I. (2010). Measuring submaximal performance parameters to monitor fatigue and predict cycling performance: A case study of a world-class cyclo-cross cyclist. European Journal of Applied Physiology, 108(1), 183–190. https://doi.org/10.1007/s00421-009-1291-3
Lamberts, R. P., Swart, J., Noakes, T. D., & Lambert, M. I. (2011). A novel submaximal cycle test to monitor fatigue and predict cycling performance. British Journal of Sports Medicine, 45(10), 797–804. https://doi.org/10.1136/bjsm.2009.061325
Lamberts, R. P., van Erp, T., Javaloyes, A., Eken, M. M., Langerak, N. G., & Tam, N. (2024). Reliability of recovery heart rate variability measurements as part of the Lamberts Submaximal Cycle Test and the relationship with training status in trained to elite cyclists. European Journal of Applied Physiology, 124(6), 1659–1668. https://doi.org/10.1007/s00421-023-05385-z
Le Meur, Y., Pichon, A., Schaal, K., Schmitt, L., Louis, J., Gueneron, J., Vidal, P. P., & Hausswirth, C. (2013). Evidence of Parasympathetic Hyperactivity in Functionally Overreached Athletes. Medicine & Science in Sports & Exercise, 45(11), 2061–2071. https://doi.org/10.1249/mss.0b013e3182980125
Lipka, A., Luthardt, C., Tognaccioli, T., Cairo, B., & Abreu, R. M. de. (2025). Heart rate variability and overtraining in soccer players: A systematic review. Physiological Reports, 13(10). https://doi.org/10.14814/phy2.70357
Lundstrom, C. J., Biltz, G. R., Uithoven, K. E., & Snyder, E. M. (2024). Effects of marathon training on heart rate variability during submaximal running: A comparison of analysis techniques. Sport Sciences for Health, 20(1), 47–54. https://doi.org/10.1007/s11332-023-01062-y
Lundstrom, C. J., Foreman, N. A., & Biltz, G. (2023). Practices and Applications of Heart Rate Variability Monitoring in Endurance Athletes. International Journal of Sports Medicine, 44(01), 9–19. https://doi.org/10.1055/a-1864-9726
Manresa‐Rocamora, A., Flatt, A. A., Casanova‐Lizón, A., Ballester‐Ferrer, J. A., Sarabia, J. M., Vera‐Garcia, F. J., & Moya‐Ramón, M. (2021). Heart rate‐based indices to detect parasympathetic hyperactivity in functionally overreached athletes. A meta‐analysis. Scandinavian Journal of Medicine & Science in Sports, 31(6), 1164–1182. https://doi.org/10.1111/sms.13932
Manser, P., & de Bruin, E. D. (2024). Test-retest reliability and validity of vagally-mediated heart rate variability to monitor internal training load in older adults: A within-subjects (repeated-measures) randomized study. BMC Sports Science, Medicine and Rehabilitation, 16(1). https://doi.org/10.1186/s13102-024-00929-y
Manzi, V., Castagna, C., Padua, E., Lombardo, M., D’Ottavio, S., Massaro, M., Volterrani, M., & Iellamo, F. (2009). Dose-response relationship of autonomic nervous system responses to individualized training impulse in marathon runners. American Journal of Physiology-Heart and Circulatory Physiology, 296(6), H1733–H1740. https://doi.org/10.1152/ajpheart.00054.2009
McGuigan, H., Hassmén, P., Rosic, N., & Stevens, C. J. (2020). Training monitoring methods used in the field by coaches and practitioners: A systematic review. International Journal of Sports Science & Coaching, 15(3), 439–451. https://doi.org/10.1177/1747954120913172
Medeiros, A. R., Leicht, A. S., Michael, S., & Boullosa, D. (2021). Weekly vagal modulations and their associations with physical fitness and physical activity. European Journal of Sport Science, 21(9), 1326–1336. https://doi.org/10.1080/17461391.2020.1838619
Medellín Ruiz, J. P., Rubio-Arias, J. Á., Clemente-Suarez, V. J., & Ramos-Campo, D. J. (2020). Effectiveness of Training Prescription Guided by Heart Rate Variability Versus Predefined Training for Physiological and Aerobic Performance Improvements: A Systematic Review and Meta-Analysis. Applied Sciences, 10(23), 8532. https://doi.org/10.3390/app10238532
Michael, S., Graham, K. S., & Davis, G. M. (2017). Cardiac Autonomic Responses during Exercise and Post-exercise Recovery Using Heart Rate Variability and Systolic Time Intervals—A Review. Frontiers in Physiology, 8, 301. https://doi.org/10.3389/fphys.2017.00301
Mirto, M., Filipas, L., Altini, M., Codella, R., & Meloni, A. (2024). Heart Rate Variability in Professional and Semiprofessional Soccer: A Scoping Review. Scandinavian Journal of Medicine & Science in Sports, 34(6). e14673. https://doi.org/10.1111/sms.14673
Mishica, C., Kyröläinen, H., Hynynen, E., Nummela, A., Holmberg, H.-C., & Linnamo, V. (2022). Evaluation of nocturnal vs. Morning measures of heart rate indices in young athletes. PLOS ONE, 17(1), e0262333. https://doi.org/10.1371/journal.pone.0262333
Moore, L., Isoard-Gautheur, S., & Gustafsson, H. (2025). Psychophysiological markers of athlete burnout: a call to arms. International journal of sports medicine, 46(2), 69–78. https://doi.org/10.1055/a-2433-3930
Nummela, A., Hynynen, E., Kaikkonen, P., & Rusko, H. (2010). Endurance Performance and Nocturnal HRV Indices. International Journal of Sports Medicine, 31(03), 154–159. https://doi.org/10.1055/s-0029-1243221
Nuuttila, O.-P., Kyröläinen, H., Kokkonen, V.-P., & Uusitalo, A. (2024). Morning versus Nocturnal Heart Rate and Heart Rate Variability Responses to Intensified Training in Recreational Runners. Sports Medicine - Open, 10(1), 120. https://doi.org/10.1186/s40798-02
Nuuttila, O.-P., Nummela, A., Korhonen, E., Häkkinen, K., & Kyröläinen, H. (2022). Individualized Endurance Training Based on Recovery and Training Status in Recreational Runners. Medicine & Science in Sports & Exercise, 54(10), 1690–1701. https://doi.org/10.1249/mss.0000000000002968
Nuuttila, O.-P., Uusitalo, A., Kokkonen, V., Weerarathna, N., & Kyröläinen, H. (2024). Monitoring fatigue state with heart rate‐based and subjective methods during intensified training in recreational runners. European Journal of Sport Science, 24(7), 857–869. https://doi.org/10.1002/ejsc.12115
Plews, D. J., Laursen, P. B., Kilding, A. E., & Buchheit, M. (2012). Heart rate variability in elite triathletes, is variation in variability the key to effective training? A case comparison. European Journal of Applied Physiology, 112(11), 3729–3741. https://doi.org/10.1007/s00421-012-2354-4
Plews, D. J., Laursen, P. B., Kilding, A. E., & Buchheit, M. (2013). Evaluating Training Adaptation With Heart-Rate Measures: A Methodological Comparison. International Journal of Sports Physiology and Performance, 8(6), 688–691. https://doi.org/10.1123/ijspp.8.6.688
Plews, D. J., Laursen, P. B., Le Meur, Y., Hausswirth, C., Kilding, A. E., & Buchheit, M. (2014). Monitoring training with heart rate-variability: how much compliance is needed for valid assessment?. International journal of sports physiology and performance, 9(5), 783–790. https://doi.org/10.1123/ijspp.2013-0455
Plews, D. J., Laursen, P. B., Stanley, J., Kilding, A. E., & Buchheit, M. (2013). Training adaptation and heart rate variability in elite endurance athletes: opening the door to effective monitoring. Sports medicine (Auckland, N.Z.), 43(9), 773–781. https://doi.org/10.1007/s40279-013-0071-8
Rabbani, A., Clemente, F. M., Kargarfard, M., & Chamari, K. (2019). Match Fatigue Time-Course Assessment Over Four Days: Usefulness of the Hooper Index and Heart Rate Variability in Professional Soccer Players. Frontiers in Physiology, 10, 109. https://doi.org/10.3389/fphys.2019.00109
Rabbani, A., Kargarfard, M., & Twist, C. (2018). Reliability and Validity of a Submaximal Warm-up Test for Monitoring Training Status in Professional Soccer Players. Journal of Strength and Conditioning Research, 32(2), 326–333. https://doi.org/10.1519/jsc.0000000000002335
Roete, A. J., Elferink-Gemser, M. T., Otter, R. T. A., Stoter, I. K., & Lamberts, R. P. (2021). A Systematic Review on Markers of Functional Overreaching in Endurance Athletes. International Journal of Sports Physiology and Performance, 16(8), 1065–1073. https://doi.org/10.1123/ijspp.2021-0024
Rothschild, J. A., Stewart, T., Kilding, A. E., & Plews, D. J. (2024). Predicting daily recovery during long-term endurance training using machine learning analysis. European Journal of Applied Physiology, 124(11), 3279–3290. https://doi.org/10.1007/s00421-024-05530-2
Ruf, L., Altmann, S., Härtel, S., Skorski, S., Drust, B., & Meyer, T. (2023). Psychophysiological Responses to a Preseason Training Camp in High-Level Youth Soccer Players. International Journal of Sports Physiology and Performance, 18(1), 18–26. https://doi.org/10.1123/ijspp.2022-0179
Ruf, L., Drust, B., Ehmann, P., Skorski, S., & Meyer, T. (2022). Are Measurement Instruments Responsive to Assess Acute Responses to Load in High-Level Youth Soccer Players? Frontiers in Sports and Active Living, 4. https://doi.org/10.3389/fspor.2022.879858
Sanchez, R., Nieto, C., Leppe, J., Gabbett, T., & Besomi, M. (2025). Associations between training load, heart rate variability, perceptual fatigue, sleep, and injury in endurance athletes during a 12-week training mesocycle. International Journal of Sports Science & Coaching, 20(5), 1918–1 928. https://doi.org/10.1177/17479541251335613
Sandercock, G. R. H., & Brodie, D. A. (2006). The use of heart rate variability measures to assess autonomic control during exercise. Scandinavian Journal of Medicine & Science in Sports, 16(5), 302–313. https://doi.org/10.1111/j.1600-0838.2006.00556.x
Schaffarczyk, M., Rogers, B., Reer, R., & Gronwald, T. (2022). Fractal correlation properties of HRV as a noninvasive biomarker to assess the physiological status of triathletes during simulated warm-up sessions at low exercise intensity: A pilot study. BMC Sports Science, Medicine and Rehabilitation, 14(1), 210. https://doi.org/10.1186/s13102-022-00596-x
Schimpchen, J., Correia, P. F., & Meyer, T. (2023). Minimally Invasive Ways to Monitor Changes in Cardiocirculatory Fitness in Running-based Sports: A Systematic Review. International journal of sports medicine, 44(2), 95–107. https://doi.org/10.1055/a-1925-7468
Schmitt, L., Bouthiaux, S., & Millet, G. P. (2021). Eleven Years’ Monitoring of the World’s Most Successful Male Biathlete of the Last Decade. International Journal of Sports Physiology and Performance, 16(6), 900–905. https://doi.org/10.1123/ijspp.2020-0148
Schmitt, L., Regnard, J., Parmentier, A., Mauny, F., Mourot, L., Coulmy, N., & Millet, G. (2015). Typology of “Fatigue” by Heart Rate Variability Analysis in Elite Nordic-skiers. International Journal of Sports Medicine, 36(12), 999–1007. https://doi.org/10.1055/s-0035-1548885
Schneider, C., Hanakam, F., Wiewelhove, T., Döweling, A., Kellmann, M., Meyer, T., Pfeiffer, M., & Ferrauti, A. (2018). Heart Rate Monitoring in Team Sports—A Conceptual Framework for Contextualizing Heart Rate Measures for Training and Recovery Prescription. Frontiers in Physiology, 9. https://doi.org/10.3389/fphys.2018.00639
Schneider, C., Wiewelhove, T., Raeder, C., Flatt, A. A., Hoos, O., Hottenrott, L., Schumbera, O., Kellmann, M., Meyer, T., Pfeiffer, M., & Ferrauti, A. (2019). Heart Rate Variability Monitoring During Strength and High-Intensity Interval Training Overload Microcycles. Frontiers in Physiology, 10, 582. https://doi.org/10.3389/fphys.2019.00582
Seiler, S., Haugen, O., & Kuffel, E. (2007). Autonomic Recovery after Exercise in Trained Athletes: Intensity and Duration Effects. Medicine & Science in Sports & Exercise, 39(8), 1366–1373. https://doi.org/10.1249/mss.0b013e318060f17d
Shushan, T., Lovell, R., Buchheit, M., Scott, T. J., Barrett, S., Norris, D., & McLaren, S. J. (2023). Submaximal Fitness Test in Team Sports: A Systematic Review and Meta-Analysis of Exercise Heart Rate Measurement Properties. Sports Medicine - Open, 9(1). https://doi.org/10.1186/s40798-023-00564-w
Shushan, T., Lovell, R., McLaren, S. J., Buchheit, M., Dello Iacono, A., Arguedas-Soley, A., & Norris, D. (2024). Assessing criterion and longitudinal validity of submaximal heart rate indices as measures of cardiorespiratory fitness: A preliminary study in football. Journal of Science and Medicine in Sport, 27(8), 565–571. https://doi.org/10.1016/j.jsams.2024.04.006
Shushan, T., McLaren, S. J., Buchheit, M., Scott, T. J., Barrett, S., & Lovell, R. (2022). Submaximal Fitness Tests in Team Sports: A Theoretical Framework for Evaluating Physiological State. Sports Medicine, 52(11), 2605–2626. https://doi.org/10.1007/s40279-022-01712-0
Siegl, A., M. Kösel, E., Tam, N., Koschnick, S., Langerak, N., Skorski, S., Meyer, T., & Lamberts, R. (2017). Submaximal Markers of Fatigue and Overreaching; Implications for Monitoring Athletes. International Journal of Sports Medicine, 38(09), 675–682. https://doi.org/10.1055/s-0043-110226
Stanley, J., Peake, J. M., & Buchheit, M. (2013). Cardiac parasympathetic reactivation following exercise: implications for training prescription. Sports Medicine (Auckland, N.Z.), 43(12), 1259–1277. https://doi.org/10.1007/s40279-013-0083-4
Stone, J. D., Ulman, H. K., Tran, K., Thompson, A. G., Halter, M. D., Ramadan, J. H., Stephenson, M., Finomore, V. S., Jr, Galster, S. M., Rezai, A. R., & Hagen, J. A. (2021). Assessing the Accuracy of Popular Commercial Technologies That Measure Resting Heart Rate and Heart Rate Variability. Frontiers in Sports and Active Living, 3, 585870. https://doi.org/10.3389/fspor.2021.585870
Thorpe, R. T., Atkinson, G., Drust, B., & Gregson, W. (2017). Monitoring Fatigue Status in Elite Team-Sport Athletes: Implications for Practice. International Journal of Sports Physiology and Performance, 12(s2), S2-27-S2-34. https://doi.org/10.1123/ijspp.2016-0434
Vesterinen, V., Nummela, A., Äyrämö, S., Laine, T., Hynynen, E., Mikkola, J., & Häkkinen, K. (2016). Monitoring Training Adaptation With a Submaximal Running Test Under Field Conditions. International Journal of Sports Physiology and Performance, 11(3), 393–399. https://doi.org/10.1123/ijspp.2015-0366
Vesterinen, V., Nummela, A., Heikura, I., Laine, T., Hynynen, E., Botella, J., & Häkkinen, K. (2016). Individual Endurance Training Prescription with Heart Rate Variability. Medicine & Science in Sports & Exercise, 48(7), 1347–1354. https://doi.org/10.1249/mss.0000000000000910
Vesterinen, V., Nummela, A., Laine, T., Hynynen, E., Mikkola, J., & Häkkinen, K. (2017). A Submaximal Running Test With Postexercise Cardiac Autonomic and Neuromuscular Function in Monitoring Endurance Training Adaptation. Journal of Strength and Conditioning Research, 31(1), 233–243. https://doi.org/10.1519/jsc.0000000000001458
Zacher, J., Branahl, A., Predel, H.-G., & Laborde, S. (2023). Effects of Covid-19 on the autonomic nervous system in elite athletes assessed by heart rate variability. Sport Sciences for Health, 19(4), 1269–1280. https://doi.org/10.1007/s11332-023-01067-7
Zignoli, A., Plews, D. J., Laursen, P. B., & Buchheit, M. (2024). AI-assisted HRV monitoring: Enhancing training load response and decision-making. Sport Performance & Science Reports, Nov, 241, v1.
Basics and application of orthostatic testing:
Barantke, M., Krauss, T., Ortak, J., Lieb, W., Reppel, M., Burgdorf, C., Pramstaller, P. P., Schunkert, H., & Bonnemeier, H. (2008). Effects of Gender and Aging on Differential Autonomic Responses to Orthostatic Maneuvers. Journal of Cardiovascular Electrophysiology, 19(12), 1296–1303. https://doi.org/10.1111/j.1540-8167.2008.01257.x
Barrero, A., Le Cunuder, A., Carrault, G., Carré, F., Schnell, F., & Le Douairon Lahaye, S. (2020). Modeling Stress-Recovery Status Through Heart Rate Changes Along a Cycling Grand Tour. Frontiers in Neuroscience, 14. https://doi.org/10.3389/fnins.2020.576308
Barrero, A., Schnell, F., Carrault, G., Kervio, G., Matelot, D., Carré, F., & Le Douairon Lahaye, S. (2019). Daily fatigue-recovery balance monitoring with heart rate variability in well-trained female cyclists on the Tour de France circuit. PLOS ONE, 14(3), e0213472. https://doi.org/10.1371/journal.pone.0213472
Bellenger, C. R., Fuller, J. T., Thomson, R. L., Davison, K., Robertson, E. Y., & Buckley, J. D. (2016). Monitoring Athletic Training Status Through Autonomic Heart Rate Regulation: A Systematic Review and Meta-Analysis. Sports Medicine, 46(10), 1461–1486. https://doi.org/10.1007/s40279-016-0484-2
Bellenger, C. R., Karavirta, L., Thomson, R. L., Robertson, E. Y., Davison, K., & Buckley, J. D. (2016). Contextualizing Parasympathetic Hyperactivity in Functionally Overreached Athletes With Perceptions of Training Tolerance. International Journal of Sports Physiology and Performance, 11(5), 685–692. https://doi.org/10.1123/ijspp.2015-0495
Bourdillon, N., Bellenoue, S., Schmitt, L., & Millet, G. P. (2024). Daily cardiac autonomic responses during the Tour de France in a male professional cyclist. Frontiers in Neuroscience, 17. https://doi.org/10.3389/fnins.2023.1221957
Bourdillon, N., Schmitt, L., Yazdani, S., Vesin, J.-M., & Millet, G. P. (2017). Minimal Window Duration for Accurate HRV Recording in Athletes. Frontiers in Neuroscience, 11. https://doi.org/10.3389/fnins.2017.00456
Buchheit, M., Al Haddad, H., Laursen, P. B., & Ahmaidi, S. (2009). Effect of body posture on postexercise parasympathetic reactivation in men. Experimental Physiology, 94(7), 795–804. https://doi.org/10.1113/expphysiol.2009.048041
Buchheit, M., Chivot, A., Parouty, J., Mercier, D., Al Haddad, H., Laursen, P. B., & Ahmaidi, S. (2010). Monitoring endurance running performance using cardiac parasympathetic function. European Journal of Applied Physiology, 108(6), 1153–1167. https://doi.org/10.1007/s00421-009-1317-x
Buchheit, M., Simon, C., Piquard, F., Ehrhart, J., & Brandenberger, G. (2004). Effects of increased training load on vagal-related indexes of heart rate variability: A novel sleep approach. American Journal of Physiology-Heart and Circulatory Physiology, 287(6), H2813–H2818. https://doi.org/10.1152/ajpheart.00490.2004
Butler, G. C., Yamamoto, Y., & Hughson, R. L. (1994). Heart Rate Variability to Monitor Autonomic Nervous System Activity During Orthostatic Stress. The Journal of Clinical Pharmacology, 34(6), 558–562. https://doi.org/10.1002/j.1552-4604.1994.tb02007.x
Butler, G. C., Yamamoto, Y., Xing, H. C., Northey, D. R., & Hughson, R. L. (1993). Heart rate variability and fractal dimension during orthostatic challenges. Journal of Applied Physiology, 75(6), 2602–2612. https://doi.org/10.1152/jappl.1993.75.6.2602
Carnethon, M. R., Liao, D., Evans, G. W., Cascio, W. E., Chambless, L. E., & Heiss, G. (2002). Correlates of the shift in heart rate variability with an active postural change in a healthy population sample: The Atherosclerosis Risk In Communities study. American Heart Journal, 143(5), 808–813. https://doi.org/10.1067/mhj.2002.121928
da Cruz, C. J. G., Porto, L. G. G., da Silva Rolim, P., de Souza Pires, D., Garcia, G. L., & Molina, G. E. (2019). Impact of heart rate on reproducibility of heart rate variability analysis in the supine and standing positions in healthy men. Clinics, 74, e806. https://doi.org/10.6061/clinics/2019/e806
da Silva, V. P., de Oliveira, N. A., Silveira, H., Mello, R. G. T., & Deslandes, A. C. (2015). Heart Rate Variability Indexes as a Marker of Chronic Adaptation in Athletes: A Systematic Review. Annals of Noninvasive Electrocardiology, 20(2), 108–118. https://doi.org/10.1111/anec.12237
Dambrink, J. H. A., & Wieling, W. (1987). Circulatory response to postural change in healthy male subjects in relation to age. Clinical Science, 72(3), 335–341. https://doi.org/10.1042/cs0720335
Dantas, E. M., Gonçalves, C. P., Silva, A. B. T., Rodrigues, S. L., Ramos, M. S., Andreão, R. V., Pimentel, E. B., Lunz, W., & Mill, J. G. (2010). Reproducibility of heart rate variability parameters measured in healthy subjects at rest and after a postural change maneuver. Brazilian Journal of Medical and Biological Research, 43(10), 982–988. https://doi.org/10.1590/s0100-879x2010007500101
de Souza, A. C., Cisternas, J. R., de Abreu, L., Roque, A. L., Monteiro, C. B., Adami, F., Vanderlei, L. C. M., Sousa, F. H., Ferreira, L. L., & Valenti, V. E. (2014). Fractal correlation property of heart rate variability in response to the postural change maneuver in healthy women. International Archives of Medicine, 7(1), 25. https://doi.org/10.1186/1755-7682-7-25
Ewing, D. J., Campbell, I. W., Murray, A., Neilson, J. M., & Clarke, B. F. (1978). Immediate heart-rate response to standing: Simple test for autonomic neuropathy in diabetes. BMJ, 1(6106), 145–147. https://doi.org/10.1136/bmj.1.6106.145
Ewing, D. J., Hume, L., Campbell, I. W., Murray, A., Neilson, J. M., & Clarke, B. F. (1980). Autonomic mechanisms in the initial heart rate response to standing. Journal of Applied Physiology, 49(5), 809–814. https://doi.org/10.1152/jappl.1980.49.5.809
Fedorowski, A., Sheldon, R., & Sutton, R. (2023). Tilt testing evolves: Faster and still accurate. European Heart Journal, 44(27), 2480–2482. https://doi.org/10.1093/eurheartj/ehad359
Finucane, C., van Wijnen, V. K., Fan, C. W., Soraghan, C., Byrne, L., Westerhof, B. E., Freeman, R., Fedorowski, A., Harms, M. P. M., Wieling, W., & Kenny, R. (2019). A practical guide to active stand testing and analysis using continuous beat-to-beat non-invasive blood pressure monitoring. Clinical Autonomic Research, 29(4), 427–441. https://doi.org/10.1007/s10286-019-00606-y
Freeman, R. (2006). Assessment of cardiovascular autonomic function. Clinical Neurophysiology, 117(4), 716–730. https://doi.org/10.1016/j.clinph.2005.09.027
Freeman, R., Wieling, W., Axelrod, F. B., Benditt, D. G., Benarroch, E., Biaggioni, I., Cheshire, W. P., Chelimsky, T., Cortelli, P., Gibbons, C. H., Goldstein, D. S., Hainsworth, R., Hilz, M. J., Jacob, G., Kaufmann, H., Jordan, J., Lipsitz, L. A., Levine, B. D., Low, P. A., … van Dijk, J. G. (2011). Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Clinical Autonomic Research, 21(2), 69–72. https://doi.org/10.1007/s10286-011-0119-5
Gilder, M., & Ramsbottom, R. (2008). Change in heart rate variability following orthostasis relates to volume of exercise in healthy women. Autonomic Neuroscience, 143(1–2), 73–76. https://doi.org/10.1016/j.autneu.2008.06.002
Gonçalves, T. R., Farinatti, P. de T. V., Gurgel, J. L., & da Silva Soares, P. P. (2015). Correlation Between Cardiac Autonomic Modulation in Response to Orthostatic Stress and Indicators of Quality of Life, Physical Capacity, and Physical Activity in Healthy Individuals. Journal of Strength and Conditioning Research, 29(5), 1415–1421. https://doi.org/10.1519/jsc.0000000000000769
Grant, C. C., Clark, J. R., van Rensburg, D. C. J., & Viljoen, M. (2009). Relationship between exercise capacity and heart rate variability: Supine and in response to an orthostatic stressor. Autonomic Neuroscience, 151(2), 186–188. https://doi.org/10.1016/j.autneu.2009.08.015
Grant, C. C., Murray, C., Janse van Rensburg, D. C., & Fletcher, L. (2013). A comparison between heart rate and heart rate variability as indicators of cardiac health and fitness. Frontiers in Physiology, 4, 337. https://doi.org/10.3389/fphys.2013.00337
Grant, C. C., Viljoen, M., Janse van Rensburg, D. C., & Wood, P. S. (2012). Heart Rate Variability Assessment of the Effect of Physical Training on Autonomic Cardiac Control. Annals of Noninvasive Electrocardiology, 17(3), 219–229. https://doi.org/10.1111/j.1542-474x.2012.00511.x
Gratze, G., Rudnicki, R., Urban, W., Mayer, H., Schlögl, A., & Skrabal, F. (2005). Hemodynamic and autonomic changes induced by Ironman: Prediction of competition time by blood pressure variability. Journal of Applied Physiology, 99(5), 1728–1735. https://doi.org/10.1152/japplphysiol.00487.2005
Gronwald, T., Schaffarczyk, M., & Hoos, O. (2024). Orthostatic testing for heart rate and heart rate variability monitoring in exercise science and practice. European Journal of Applied Physiology, 124(12), 3495–3510. https://doi.org/10.1007/s00421-024-05601-4
Hedelin, R., Wiklund, U., Bjerle, P., & Henriksson-Larsén, K. (2000). Cardiac autonomic imbalance in an overtrained athlete. Medicine & Science in Sports & Exercise, 32(9), 1531–1533. https://doi.org/10.1097/00005768-200009000-00001
Holmes, C. J., Fedewa, M. V., Dobbs, W. C., Liu, Y., Flatt, A. A., Nakamura, F. Y., & Esco, M. R. (2020). The effects of different body positions on the accuracy of ultra-short-term heart rate variability indexes. The Journal of High Technology Management Research, 31(1), 100375. https://doi.org/10.1016/j.hitech.2020.100375
Hottenrott K. & Hoos O. (2017). Heart rate variability analysis in exercise physiology. In: H. Jelinek, A. Khandoker, & D. Cornforth (Eds.), ECG time series analysis: Engineering to medicine (pp. 245–257), London, UK: CRC Press.
Hottenrott, K. & Gronwald, T. (2014). Bedeutung der Herzfrequenzvariabilität für die Regenerationssteuerung. Leistungssport, 44(5), 9-13.
Hottenrott, L., Gronwald, T., Hottenrott, K., Wiewelhove, T., & Ferrauti, A. (2021). Utilizing Heart Rate Variability for Coaching Athletes During and After Viral Infection: A Case Report in an Elite Endurance Athlete. Frontiers in Sports and Active Living, 3, 612782. https://doi.org/10.3389/fspor.2021.612782
Hynynen, E., Konttinen, N., Kinnunen, U., Kyröläinen, H., & Rusko, H. (2011). The incidence of stress symptoms and heart rate variability during sleep and orthostatic test. European Journal of Applied Physiology, 111(5), 733–741. https://doi.org/10.1007/s00421-010-1698-x
Hynynen, E., Uusitalo, A., Konttinen, N., & Rusko, H. (2006). Heart Rate Variability during Night Sleep and after Awakening in Overtrained Athletes. Medicine & Science in Sports & Exercise, 38(2), 313–317. https://doi.org/10.1249/01.mss.0000184631.27641.b5
Hynynen, E., Uusitalo, A., Konttinen, N., & Rusko, H. (2008). Cardiac Autonomic Responses to Standing Up and Cognitive Task in Overtrained Athletes. International Journal of Sports Medicine, 29(7), 552–558. https://doi.org/10.1055/s-2007-989286
Janssen, M. J. A., de Bie, J., Swenne, C. A., & Oudhof, J. (1993). Supine and standing sympathovagal balance in athletes and controls. European Journal of Applied Physiology and Occupational Physiology, 67(2), 164–167. https://doi.org/10.1007/bf00376661
Koelwyn, G. J., Wong, L. E., Kennedy, M. D., & Eves, N. D. (2013). The effect of hypoxia and exercise on heart rate variability, immune response, and orthostatic stress. Scandinavian Journal of Medicine & Science in Sports, 23(1). https://doi.org/10.1111/sms.12003
Kowalewski, M. A., & Urban, M. (2004). Short- and long-term reproducibility of autonomic measures in supine and standing positions. Clinical Science, 106(1), 61–66. https://doi.org/10.1042/cs20030119
Le Meur, Y., Pichon, A., Schaal, K., Schmitt, L., Louis, J., Gueneron, J., Vidal, P. P., & Hausswirth, C. (2013). Evidence of Parasympathetic Hyperactivity in Functionally Overreached Athletes. Medicine & Science in Sports & Exercise, 45(11), 2061–2071. https://doi.org/10.1249/mss.0b013e3182980125
Lin, L. L.-C., Chen, Y.-J., Lin, T.-Y., & Weng, T.-C. (2022). Effects of Resistance Training Intensity on Heart Rate Variability at Rest and in Response to Orthostasis in Middle-Aged and Older Adults. International Journal of Environmental Research and Public Health, 19(17), 10579. https://doi.org/10.3390/ijerph191710579
Lipsitz, L. A., Mietus, J., Moody, G. B., & Goldberger, A. L. (1990). Spectral characteristics of heart rate variability before and during postural tilt. Relations to aging and risk of syncope. Circulation, 81(6), 1803–1810. https://doi.org/10.1161/01.cir.81.6.1803
Lucini, D., Norbiato, G., Clerici, M., & Pagani, M. (o. J.). Hemodynamic and Autonomic Adjustments to Real Life Stress Conditions in Humans.
Maggioni, M. A., Rundfeldt, L. C., Gunga, H.-C., Joerres, M., Merati, G., & Steinach, M. (2020). The Advantage of Supine and Standing Heart Rate Variability Analysis to Assess Training Status and Performance in a Walking Ultramarathon. Frontiers in Physiology, 11, 731. https://doi.org/10.3389/fphys.2020.00731
Manresa‐Rocamora, A., Flatt, A. A., Casanova‐Lizón, A., Ballester‐Ferrer, J. A., Sarabia, J. M., Vera‐Garcia, F. J., & Moya‐Ramón, M. (2021). Heart rate‐based indices to detect parasympathetic hyperactivity in functionally overreached athletes. A meta‐analysis. Scandinavian Journal of Medicine & Science in Sports, 31(6), 1164–1182. https://doi.org/10.1111/sms.13932
Manser, P., Thalmann, M., Adcock, M., Knols, R. H., & de Bruin, E. D. (2021). Can Reactivity of Heart Rate Variability Be a Potential Biomarker and Monitoring Tool to Promote Healthy Aging? A Systematic Review With Meta-Analyses. Frontiers in Physiology, 12, 686129. https://doi.org/10.3389/fphys.2021.686129
McCrory, C., Berkman, L. F., Nolan, H., O’Leary, N., Foley, M., & Kenny, R. A. (2016). Speed of Heart Rate Recovery in Response to Orthostatic Challenge. Circulation Research, 119(5), 666–675. https://doi.org/10.1161/circresaha.116.308577
Mellingsæter, M. R., Wyller, T. B., Ranhoff, A. H., & Wyller, V. B. (2015). Fit elderly men can also stand: Orthostatic tolerance and autonomic cardiovascular control in elderly endurance athletes. Aging Clinical and Experimental Research, 27(4), 499–505. https://doi.org/10.1007/s40520-014-0303-2
Montano, N., Ruscone, T. G., Porta, A., Lombardi, F., Pagani, M., & Malliani, A. (1994). Power spectrum analysis of heart rate variability to assess the changes in sympathovagal balance during graded orthostatic tilt. Circulation, 90(4), 1826–1831. https://doi.org/10.1161/01.cir.90.4.1826
Mourot, L., Bouhaddi, M., Perrey, S., Cappelle, S., Henriet, M., Wolf, J., Rouillon, J., & Regnard, J. (2004). Decrease in heart rate variability with overtraining: Assessment by the Poincaré plot analysis. Clinical Physiology and Functional Imaging, 24(1), 10–18. https://doi.org/10.1046/j.1475-0961.2003.00523.x
Murrell, C., Wilson, L., Cotter, J. D., Lucas, S., Ogoh, S., George, K., & Ainslie, P. N. (2007). Alterations in autonomic function and cerebral hemodynamics to orthostatic challenge following a mountain marathon. Journal of Applied Physiology, 103(1), 88–96. https://doi.org/10.1152/japplphysiol.01396.2006
Ravé, G., & Fortrat, J.-O. (2016). Heart rate variability in the standing position reflects training adaptation in professional soccer players. European Journal of Applied Physiology, 116(8), 1575–1582. https://doi.org/10.1007/s00421-016-3416-9
Schäfer, D., Nil, M., Herzig, D., Eser, P., Saner, H., & Wilhelm, M. (2015). Good reproducibility of heart rate variability after orthostatic challenge in patients with a history of acute coronary syndrome. Journal of Electrocardiology, 48(4), 696–702. https://doi.org/10.1016/j.jelectrocard.2015.04.004
Schmitt, L., Regnard, J., & Millet, G. P. (2015). Monitoring Fatigue Status with HRV Measures in Elite Athletes: An Avenue Beyond RMSSD? Frontiers in Physiology, 6, 343. https://doi.org/10.3389/fphys.2015.00343
Schmitt, L., Regnard, J., Desmarets, M., Mauny, F., Mourot, L., Fouillot, J.-P., Coulmy, N., & Millet, G. (2013). Fatigue Shifts and Scatters Heart Rate Variability in Elite Endurance Athletes. PLoS ONE, 8(8), e71588. https://doi.org/10.1371/journal.pone.0071588
Schmitt, L., Regnard, J., Parmentier, A., Mauny, F., Mourot, L., Coulmy, N., & Millet, G. (2015). Typology of “Fatigue” by Heart Rate Variability Analysis in Elite Nordic-skiers. International Journal of Sports Medicine, 36(12), 999–1007. https://doi.org/10.1055/s-0035-1548885
Schneider, C., Hanakam, F., Wiewelhove, T., Döweling, A., Kellmann, M., Meyer, T., Pfeiffer, M., & Ferrauti, A. (2018). Heart Rate Monitoring in Team Sports—A Conceptual Framework for Contextualizing Heart Rate Measures for Training and Recovery Prescription. Frontiers in Physiology, 9. https://doi.org/10.3389/fphys.2018.00639
Schneider, C., Wiewelhove, T., Raeder, C., Flatt, A. A., Hoos, O., Hottenrott, L., Schumbera, O., Kellmann, M., Meyer, T., Pfeiffer, M., & Ferrauti, A. (2019). Heart Rate Variability Monitoring During Strength and High-Intensity Interval Training Overload Microcycles. Frontiers in Physiology, 10. https://doi.org/10.3389/fphys.2019.00582
Soloveva, A., Fedorova, D., Villevalde, S., Zvartau, N., Mareev, Y., Sitnikova, M., Shlyakhto, E., & Fudim, M. (2020). Addressing Orthostatic Hypotension in Heart Failure: Pathophysiology, Clinical Implications and Perspectives. Journal of Cardiovascular Translational Research, 13(4), 549–569. https://doi.org/10.1007/s12265-020-10044-1
Stewart, J. M. (2012). Mechanisms of sympathetic regulation in orthostatic intolerance. Journal of Applied Physiology, 113(10), 1659–1668. https://doi.org/10.1152/japplphysiol.00266.2012
Sutton, R., Fedorowski, A., Olshansky, B., Gert van Dijk, J., Abe, H., Brignole, M., de Lange, F., Kenny, R. A., Lim, P. B., Moya, A., Rosen, S. D., Russo, V., Stewart, J. M., Thijs, R. D., & Benditt, D. G. (2021). Tilt testing remains a valuable asset. European Heart Journal, 42(17), 1654–1660. https://doi.org/10.1093/eurheartj/ehab084
Swai, J., Hu, Z., Zhao, X., Rugambwa, T., & Ming, G. (2019). Heart rate and heart rate variability comparison between postural orthostatic tachycardia syndrome versus healthy participants; a systematic review and meta-analysis. BMC Cardiovascular Disorders, 19(1), 320. https://doi.org/10.1186/s12872-019-01298-y
Taylor, A. A. (1994). Autonomic Control of Cardiovascular Function: Clinical Evaluation in Health and Disease. The Journal of Clinical Pharmacology, 34(5), 363–374. https://doi.org/10.1002/j.1552-4604.1994.tb04976.x
Thayer, J. F., Yamamoto, S. S., & Brosschot, J. F. (2010). The relationship of autonomic imbalance, heart rate variability and cardiovascular disease risk factors. International Journal of Cardiology, 141(2), 122–131. https://doi.org/10.1016/j.ijcard.2009.09.543
Tulppo, M. P., Hughson, R. L., Mäkikallio, T. H., Airaksinen, K. E. J., Seppänen, T., & Huikuri, H. V. (2001). Effects of exercise and passive head-up tilt on fractal and complexity properties of heart rate dynamics. American Journal of Physiology-Heart and Circulatory Physiology, 280(3), H1081–H1087. https://doi.org/10.1152/ajpheart.2001.280.3.H1081
Uusitalo, A. L., Uusitalo, A. J., & Rusko, H. K. (2000). Heart rate and blood pressure variability during heavy training and overtraining in the female athlete. International Journal of Sports Medicine, 21(1), 45–53. https://doi.org/10.1055/s-2000-8853
van Wijnen, V. K., Finucane, C., Harms, M. P. M., Nolan, H., Freeman, R. L., Westerhof, B. E., Kenny, R. A., ter Maaten, J. C., & Wieling, W. (2017). Noninvasive beat‐to‐beat finger arterial pressure monitoring during orthostasis: A comprehensive review of normal and abnormal responses at different ages. Journal of Internal Medicine, 282(6), 468–483. https://doi.org/10.1111/joim.12636
van Zanten, S., Sutton, R., Hamrefors, V., Fedorowski, A., & de Lange, F. J. (2024). Tilt table testing, methodology and practical insights for the clinic. Clinical Physiology and Functional Imaging, 44(2), 119–130. https://doi.org/10.1111/cpf.12859
Vescovi J. D. (2019). Intra-Individual Variation of HRV during Orthostatic Challenge in Elite Male Field Hockey Players. Journal of medical systems, 43(12), 328. https://doi.org/10.1007/s10916-019-1478-z
Vinik, A. I., Maser, R. E., Mitchell, B. D., & Freeman, R. (2003). Diabetic autonomic neuropathy. Diabetes care, 26(5), 1553–1579. https://doi.org/10.2337/diacare.26.5.1553
Vybiral, T., Bryg, R. J., Maddens, M. E., & Boden, W. E. (1989). Effect of passive tilt on sympathetic and parasympathetic components of heart rate variability in normal subjects. The American Journal of Cardiology, 63(15), 1117–1120. https://doi.org/10.1016/0002-9149(89)90089-1
Young, F. L. S., & Leicht, A. S. (2011). Short-term stability of resting heart rate variability: Influence of position and gender. Applied Physiology, Nutrition, and Metabolism, 36(2), 210–218. https://doi.org/10.1139/h10-103


























